Phenotypes associated with this allele
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
|
|
|
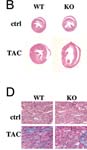
Ppargc1atm1Brsp/Ppargc1atm1Brsp mice develop dilated cardiomyopathy in response to transverse aortic constriciton (TAC)
cardiovascular system
|
|
• hearts from mutants subjected to transverse aortic constriction are more increased in weight and dilated than wild-type
|
|
|
• 2 months after transverse aortic constriction (TAC), develop profound cardiac dysfunction, with hearts becoming dilated and showing impaired ability to contract
|
|
|
• TAC leads to accelerated cardiac dysfunction, accompanied by signs of heart failure
|
immune system
growth/size/body
|
|
• hearts from mutants subjected to transverse aortic constriction are more increased in weight and dilated than wild-type
|
|
|
• 2 months after TAC, mutants are visibly emaciated and show significant drops in weight, unlike wild-type
|
|
|
• lungs are nearly quadrupled in weight 2 months after TAC as compared with only a moderate increase in wild-type TAC mice
|
homeostasis/metabolism
respiratory system
|
|
• lungs are nearly quadrupled in weight 2 months after TAC as compared with only a moderate increase in wild-type TAC mice
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
|
|
|
mortality/aging
|
|
• homozygotes die when exposed to 4oC for more than 6 hours
|
|
|
• about 50% of homozygotes die during the postnatal period
|
adipose tissue
|
|
• abundant, large lipid droplets are seen in the brown fat
|
behavior/neurological
|
|
• mice exhibit decreased sucrose consumption as compared to wild-type mice
• sucrose consumption is decreased further following administration of kynurenine (KYN)
|
|
|
• an exaggerated startle response is seen
|
|
|
• dystonic posturing is seen
|
|
|
• a 40% increase in the frequency of random movements is seen
|
|
|
• stimulus induced myoclonus is seen
|
cellular
|
|
• respiration due to mitochondrial proton leak is increased 20% in primary hepatocytes
|
homeostasis/metabolism
muscle
|
|
• dystonic posturing is seen
|
|
|
• stimulus induced myoclonus is seen
|
nervous system
|
|
• stimulus induced myoclonus is seen
|
|
|
• gliosis is seen associated with the spongiform lesions
|
|
|
• striatal neurons with reduced branches of neurites and occasional vacuoles within the neurons are seen
|
|
|
• spongiform lesions are seen predominantly in the striatum and to a lesser extent in the cortex
• the lesions are mostly associated with the white matter
|
growth/size/body
|
|
• on a high fat diet homozygous mutants are significantly leaner compared to wild-type mice
|
liver/biliary system
|
|
• triglyceride levels are lower in the livers of fasted mutant mice
|
Allelic
Composition |
Ppargc1atm1Brsp/Ppargc1a+
|
|
Genetic
Background |
involves: 129 * FVB/N |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
|
|
|
immune system
|
N |
• mice exhibit normal serum IL6 levels
|
Allelic
Composition |
Ppargc1atm1Brsp/Ppargc1a+
|
|
Genetic
Background |
involves: 129S4/SvJae * C57BL/6 |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
|
|
|
vision/eye
|
|
• mice fed a regular diet show enlarged blood vessels in the interface between the Bruch's membrane and the choroid, with congestion and dilatation of some vessels
|
|
|
• mice fed a high-fat diet show a greater loss of fenestration in choriocapillaris endothelium
|
|
|
• mice fed a regular diet and a high-fat diet show a reduction of thickness of the inner segment layer
|
|
|
• mice fed a regular diet and a high-fat diet show a reduction of thickness of the outer segment layer
|
|
|
• mice fed a high-fat diet exhibit a thinner photoreceptor layer, indicating degeneration of this layer
• photoreceptor degeneration does not necessarily initiate concurrently in both eyes
|
|
|
• mice fed a regular diet show higher numbers of lipofuscin deposits in the retinal pigment epithelium (RPE) than wild-type mice
• mice fed a high-fat diet show a greater accumulation of lipofuscin in the cytoplasm of the RPE, basal laminar deposits, and thickening of the outer collagenous layer
• mice fed a high-fat diet show RPE retinal pigment epithelium degeneration, with disruptions or gaps and scant melanosomes in the subretinal space migrating into the outer segment
|
|
|
• mice fed a high-fat diet present age-related macular degeneration-like abnormalities in the retinal pigment epithelium and retinal morphology and function
|
|
|
• mice fed a high-fat diet show changes in the Bruch membrane, either atrophy or thickening in various regions
• mice fed a high-fat diet show accumulation of carboxymethyl lysine (CML) deposits in the thickened Bruch membrane, indicating oxidative damage
|
immune system
|
|
• lipopolysaccharide (LPS) injection induces a higher inflammatory response in the retinal pigment epithelium (RPE)/retina compared to in wild-type mice
|
cardiovascular system
|
|
• mice fed a regular diet show enlarged blood vessels in the interface between the Bruch's membrane and the choroid, with congestion and dilatation of some vessels
|
|
|
• mice fed a high-fat diet show a greater loss of fenestration in choriocapillaris endothelium
|
homeostasis/metabolism
nervous system
|
|
• mice fed a regular diet and a high-fat diet show a reduction of thickness of the inner segment layer
|
|
|
• mice fed a regular diet and a high-fat diet show a reduction of thickness of the outer segment layer
|
|
|
• mice fed a high-fat diet exhibit a thinner photoreceptor layer, indicating degeneration of this layer
• photoreceptor degeneration does not necessarily initiate concurrently in both eyes
|
pigmentation
|
|
• mice fed a regular diet show higher numbers of lipofuscin deposits in the retinal pigment epithelium (RPE) than wild-type mice
• mice fed a high-fat diet show a greater accumulation of lipofuscin in the cytoplasm of the RPE, basal laminar deposits, and thickening of the outer collagenous layer
• mice fed a high-fat diet show RPE retinal pigment epithelium degeneration, with disruptions or gaps and scant melanosomes in the subretinal space migrating into the outer segment
|
|
|
• mice fed a high-fat diet show scant melanosomes migrating into the outer segment
|
|
|
• mice fed a regular diet show higher numbers of lipofuscin deposits than wild-type mice
• mice fed a high-fat diet show a greater accumulation of lipofuscin in the cytoplasm of the RPE
|
cellular
|
|
• mice fed a high-fat diet show a greater decrease in mtDNA copy number than wild-type mice
|
|
|
• autophagy is reduced in the retinal pigment epithelium/retina
|
|
|
• mice fed a high-fat diet show a greater decrease in mitochondrial complex I activity in the retinal pigment epithelium/retina
|
|
|
• mice fed a high-fat diet show accumulation of carboxymethyl lysine deposits in the thickened Bruchs membrane, indicating oxidative damage
• mice fed a high-fat diet show increased reactive oxygen species (ROS) levels in the retinal pigment epithelium/retina
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
Ppargc1atm2Brsp mutation
(0 available);
any
Ppargc1a mutation
(47 available)
Tg(Myog-cre)1Eno mutation
(0 available)
|
|
|
homeostasis/metabolism
endocrine/exocrine glands
|
N |
• islets exhibit normal basal and glucose-stimulated insulin secretion
|
growth/size/body
|
|
• in mice fed standard chow or a high fat diet
|
|
|
• in mice fed a high fat diet
|
immune system
adipose tissue
|
|
• in mice fed standard chow or a high fat diet
|
cellular
|
|
• decreased function as measured by Cox and succinate dehydrogenase activity
|
behavior/neurological
|
|
• during the day and night in mice fed a high fat diet
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Mirc33tm1.1Wtsi mutation
(0 available);
any
Mirc33 mutation
(0 available)
Ppargc1atm1Brsp mutation
(1 available);
any
Ppargc1a mutation
(47 available)
|
|
|
mortality/aging
|
|
• mice exhibit a life span extension compared with single Mirc33tm1.1Wtsi homozygotes, with a median of 39.5 weeks compared to 30 weeks, but reduced survival compared to wild-type mice
|
cellular
|
|
• cardiac mitochondrial size is smaller
• however, cardiac mitochondrial DNA levels and mitochondrial numbers are restored to wild-type levels
|
cardiovascular system
|
N |
• mice show a reduction in systemic blood pressure compared to single Mirc33tm1.1Wtsi homozygotes
|



 Analysis Tools
Analysis Tools