| Previous | Next |
Intensive study of the phenomena associated with the acceptance and rejection of tissue transplants has led to the establishment of two principles. First, rejection depends on genetic disparity of donor and host. Second, the processes involved in rejection are immunological in nature. We shall be concerned here only with the genetic factors in transplant rejection. For information concerning the immunological phenomena the reader may consult Snell ( 1963), Billingham and Silvers ( 1963), or Russell and Monaco ( 1965).
DEFINITIONS
The vocabulary of transplantation includes certain terms that may not be generally familiar. The problem of unfamiliarity is compounded by a diversity of usages, including some that are notably inconsistent. To smooth the path of the reader we shall employ a vocabulary recently suggested ( Snell, 1964b) which essentially eliminates the inconsistencies and contradictions. The key terms are defined herewith.
Histocompatibility is an adjective used to indicate relevance to the growth or failure to grow of tissue transplants. Thus histocompatibility genes are the genes that determine susceptibility and resistance to the transplants.
Isogenic is an adjective used to indicate genetic identity of animals or tissues. The members of one inbred strain are isogenic. Isogeneic is a variant used, usually as a contrasting term to allogeneic, to indicate similarity of origin.
Coisogenic strains are strains genetically identical except for a difference at a single genetic locus. Since true coisogenicity is a theoretical ideal seldom if ever attained in practice, it is convenient to have a term applicable to those strains actually available which provide an imperfect but useful approximation to the coisogenic state. Such strains may be called congenic.
An isogenic graft or isograft is a graft between genetically identical individuals. Typically, isografts are either grafts between animals of a single highly inbred strain, between the F1 hybrids produced by crossing inbred strains, or between identical twins.
An allogeneic graft or allograft is a graft between genetically disparate individuals of the same species. More specifically, it is a graft in which the grafted tissue carries a histocompatibility allele or alleles, and hence presumably an alloantigen or alloantigens, foreign to the recipient. Homograft is also used in this sense, but because its use has led to the use of homologous with a meaning exactly opposite from the traditional one, it is not recommended.
An alloantigen or isoantigen is the heteromorphic product of a heteromorphic locus (a locus existing in two or more allelic forms), and a product such that alternative forms are antigenic in individuals lacking them. It incites an immune response when transferred within the species. Alloantigen is a new and unfamiliar term in this context. We shall use it in preference to isoantigen because it leads to a more consistent vocabulary. The type of antigenic stimulus here referred to is provided by allografts, not by isografts; by grafts between individuals that are nonisogenic, not between individuals that are isogenic. It is therefore more appropriate to speak of the active substances as alloantigens than as isoantigens.
TYPES OF TRANSPLANTS
Both transplantable tumors and various normal tissues have been used in studies of the genetics of transplantation. Nearly all early work was done with tumors, and tumors are still the instrument of choice for many studies, but increasing use is being made of normal-tissue transplants.
The great advantage of tumors is the rapidity with which transplants can be performed. One experienced person can graft 500 or more animals in a day. If either leukemias, which are easily prepared as suspensions of single cells ( Snell, 1953), or ascites tumors, which grow as suspensions of single cells in the fluid-filled peritoneal cavity, are used, an accurately measured dose of cells can be administered. In tests of histocompatibility, the tumors, whether of ascites type or in the solid form, are typically implanted subcutaneously on the flank. Intramuscular implants have also been used. Death or survival of the host is taken as the end point.
If properly employed, tumors can be sensitive indicators of histocompatibility differences. Prior immunization of the recipient by one or more injections of tissue from the donor strain is frequently used to increase the reaction against the ultimate graft. Leukemias are particularly sensitive indicators of histocompatibility differences, probably because of their especial susceptibility to antibody. Tumors are also easily stored in the frozen state (see, e.g., Hauschka et al., 1959). On the other hand tumors, since they are living cell populations, may change with successive passages from host to host and hence lose desireable characteristics; they may become infected, either as the result of careless transplantation or because of an unsuspected infection in their host; and they may not be available in a particular inbred strain or hybrid which it is desired to use as a donor. (For further information on the characteristics of transplantable tumors, see Chapter 28 or Snell, 1959).
The normal tissue most commonly used in studies of the genetics of histocompatibilty is skin. Techniques for skin grafting in mice are well established ( Billingham and Silvers, 1961), and this tissue is among the more sensitive indicators of histocompatibility differences. Transplants of ovaries into ovariectomized hosts (see, e.g., Linder, 1961) have also been used. With this tissue, growth or rejection may be determined either by the continuation of the estrus cycle, or, if ovaries are placed in the ovarian capsule, by the production of young. For purpose other than genetic studies, a great deal of use has been made of suspensions of bone marrow and the lymphoid tissues. Because the cells of such transplants become widely dispersed and are not easily distinguished from host cells, there is usually no clear end point of rejection. Cudkowicz ( Cudkowicz and Stimpfling, 1964a), however, has developed an ingenious method of measuring the growth in irradiated hosts of transplanted marrow cells by the use of a radioactive DNA precursor. This method has proved suitable for genetic studies.
FIVE LAWS OF TRANSPLANTATION
Studies which established the basic principles of the genetics of tissue transplantation were started by Little and Tyzzer ( 1916) and continued by Little, Bittner, Cloudman, and Strong. All these studies were based on the use of inbred strains of mice. These early studies were summarized in some detail in the first edition of this book ( Little, 1941). Because recent data are more broadly informative, most of the early data will be omitted here and attention concentrated instead on the conclusions drawn from them. It should also be noted that when we do discuss early data, designations of substrains will be omitted. The major substrains (e.g., DBA/1 and DBA/2) were either not established or not recognized at that time.
The results of Little and co-workers can be summarized as five laws or dicta of transplantation. These are exceptions to most of these laws, especially to the second and third. These will be discussed later. Despite the exceptions, the laws have a very general validity. The laws may be stated as follows:
1. Grafts within inbred strains are successful; or stated in more general terms, isografts succeed.
2. Grafts between inbred strains are not successful; or stated in more general terms, allografts fail.
The remaining laws apply to the results which are obtained when inbred strains are crossed to produce F1, F2, and backcross generations, and grafts exchanged between the inbred parents and animals of these generations or between animals of the hybrid generations themselves.
3. Grafts from either inbred parent strain to the F1 hybrid succeed, but grafts in the reverse direction fail.
4. Grafts from F2 or subsequent generations also grow in all F1 mice.
5. Grafts from either inbred parent strain are accepted by some members of an F2 generation, but rejected by others. Usually the rejections are much more frequent than the acceptances. The same is true of grafts made from one inbred parent strain to a backcross produced by crossing the F1 to the opposite parent strain. The proportion of rejections in this generation is, for any one given cross, donor, and type of tissue, usually higher than in the F2.
REPRESENTATIVE DATA
The key to the understanding of the genetics of tissue transplantation is law 5, which concerns the results of transplants made to F2 and backcross generations. We shall, therefore, examine in some detail representative results obtained in these two generations.
Of the many tests made with transplantable tumors, two are summarized in Table 24-1. Cloudman ( 1932) crossed strains A and DBA, raised F1, F2, and backcross (BC) generations, and implanted them with a transplantable mammary tumor indigenous to strain A. This tumor grew in all animals of strain A but, except for occasional temporary growths, failed to grow in strain DBA. It grew in all of 92 F1 mice and in all of 69 mice produced by crossing the F1 to the susceptible (strain A) parent. It grew, however, in only 10 of 116 mice of the opposite backcross and 60 of 219 F2 mice.
Amos et al. ( 1955a) ran a similar test using strains C57BL and BALB/c, and a C57BL transplantable leukemia. An interesting variation in this method was the use, in some of the mice, of an "immunizing" injection of C57BL blood 6 to 10 days prior to tumor implantation. When BC mice were unimmunized, 23 out of 28 succumbed (results not shown in table). When they were immunized, all of 39 survived. In the immunized F2, 27 out of 29 survived.
Although there have been many experiments with mice in which tumors have been transplanted from an inbred strain to its F1, F2, and BC descendants, only two comparable experiments using the more tedious techniques of skin grafting have been reported. These are of considerable interest, and both are summarized in Table 24-2. In the first, Prehn and Main ( 1958) crossed strains BALB/c and DBA/2 and grafted BALB/c skin to F2 and BC generations. Of 120 grafts made to the F2, three were still healthy at 200 days and were regarded as definitive acceptances. All other grafts were rejected, although a few persisted for 60 or more days before sloughing. Of 99 grafts to backcross mice, none survived permanently, though again there were a few late rejections. The other experiment was carried out by Barnes and Krohn ( 1957). These authors crossed strains A and CBA, and grafted skin from both parent strains to F1 and F2 mice. As expected, all grafts to the F1 were accepted. About one-fourth of all grafts to the F2 had been rejected by 12 or 15 days. The survival of these grafts was approximately that expected when grafts are exchanged between inbred strains. Many other grafts looked healthy at this time. Thereafter rejections of grafts continued to occur, and by 180 days all of 274 grafts, with one possible exception, had been rejected. For purposes of discussion, the authors treated three grafts (one of strain A skin and two of CBA skin) which showed "an autograft-like condition 100 days after grafting" as acceptances. It should be noted, however, that control autografts and isografts were still healthy after 6 months or a year.
These four experiments all showed a combination of rejections and acceptances (or at least very long survivals) in the segregating generations receiving grafts from the parent strains, but the proportion of acceptances varied considerably. Specifically, the percentage of acceptances in the four F2's were 27.4, 6.9, 2.5, and 0 (or 1.7 if 100-day survival is taken as an acceptance), and for the corresponding backcross generations, 8.6, 0 (out of 39 mice), and 0 (out of 99 mice). The last experiment did not include a backcross. The considerably lower percentage of graft survival in the backcross is notable.
GENETIC THEORY OF TRANSPLANTATION
The genetic theory of transplantation in essentially its present form was adumbrated in an early paper by Little ( 1914) and was fully described in a number of subsequent papers (see, e.g., Little, 1941). Although a considerable wealth of detail has been added, and certain minor modifications have been made, the theory developed by Little and co-workers is still fully valid in its essentials.
The key to the genetics of susceptibility and resistance to allografts must be found in the F1, F2, and BC generations. The absence of a 3:1 cross suggests multiple-factor rather than single-factor inheritance. In most multiple-factor inheritance the condition which is dominant in the F1 tends to be frequently repeated in the F2, yet the exact opposite occurs here. In the transplantation studies, susceptibility is dominant in F1, but uncommon in F2. The theory of transplantation, designed to accommodate these facts, may be stated as follows:
The growth of transplants is determined by multiple genes which may be called histocompatibility genes or by the abbreviated designation H genes. Permanent or progressive growth occurs if, and only if, all histocompatibility alleles present in the graft are also present in the host. Modern research provides an intelligible explanation of this rule in terms of the end products of histocompatibility genes. These may be presumed to be alloantigens which, when transferred to a host lacking them, have a special property of inciting an immune response. Each gene is presumed to determine a single alloantigen, a situation which has led to the dictum, "one gene, one antigen." The fact that alloantigens must be foreign to the recipient to evoke an immune response accounts for the acceptance of grafts which carry no histocompatibility alleles foreign to the host, and the rejection of grafts which do carry foreign alleles. Although this extension of the genetic theory of transplantation helps us to understand it and can be supported by numerous facts, it would take us too far afield to examine it in detail. We shall for the most part examine the theory merely as a formal exercise in genetics.
We shall represent the different histocompatibility loci by the symbols H-1, H-2, H-3, etc. We shall assume that histocompatibility genes are codominant (individually expressed in the heterozygote), and shall therefore use only the capital form for the initial letter, rather than the capital and small letters which are sometimes used to indicate the dominant and recessive states. Different alleles at one locus will be indicated by superscript small letters. Thus alleles at H-1 are represented by H-1a, H-1b, H-1c, etc., alleles at H-2 by H-2a, H-2b, H-2c, etc.
Suppose, then, that two strains, A and B, with a single histocompatibility difference are crossed. Let us suppose that the difference is at the H-2 locus. The parents, the different hybrid generations, and the expected outcome of transplants from the A parent may then be represented as shown in Table 24-3. The only animals represented in this table which lack the H-2a of the graft donor H-2b/H-2b parent, and the mice of this same homozygous H-2b genotype which comprise one-fourth of the F2 generation and one-half of the BC generation. These mice will resist the H-2a/H-2a graft; all other mice will accept it. The expected results are:
| F1 | all susceptible | |
| F2 | ¾ susceptible | |
| BC | ½ susceptible |
The expected results in F2 when the parents differ at two histocompatibility loci are shown in Figure 24-1. The expected results for a backcross are similar, except that the ratio of the four classes is 1:1:1:1 instead of 9:3:3:1. The expected proportions of susceptible and resistant mice for two factors are thus:
| F1 | all susceptible | |
| F2 | (¾)2 = 9/16 susceptible | |
| BC | (½)2 = ¼ susceptible |
Generalizing from these two examples, we may say that if inbred strains differing at n histocompatibility loci are crossed, the expected proportion of susceptible animals in subsequent generations will be:
| F1 | all susceptible | |
| F2 | (¾)n susceptible | |
| BC | (½)n susceptible |
To get a picture of what this means in terms of the actual proportion of susceptible mice for different numbers of histocompatibility factors, we may appropiately select those values of n which happen to give the best fit for the four crosses shown in Tables 24-1 and 24-2. These values are, in the order in which the crosses appear in the tables, n = 4, n = 9, n = 13, and n = 15. The corresponding expected percentages of susceptible mice in F2 and backcross generations are shown in Table 24-4.
It is apparent from these values that in a backcross, even with no more than nine histocompatibility loci segregating, large numbers of mice may have to be raised to get even a single mouse that will accept a graft from the parental strain. We refer here of course to the parental strain not used in producing the backcross. For nine factors, in fact, only about one mouse in 500 will, on the average, accept a parental graft. For an F2 with nine factors, approximately one mouse in 13 will accept a parental graft. It is not surprising therefore that in two of the three crosses in which both F2 and BC generations were raised, there were positive grafts in the F2 only.
The concordance, in any one individual experiment, between observed results for F2 and BC generations and the predictions for these same generations provides an important test of the validity of the theory. An inspection of the tables will show that the concordance is satisfactory for the four crosses summarized. We may also say that it was satisfactory for most of the early experiments in which transplantable tumors were implanted in both F2 and BC generations.
There is, however, a considerable lack of concordance when the four experiments cited are compared with respect to the indicated number of histocompatibility loci. The number ranges all the way from four to not fewer than 15. Sometimes the same cross challenged with different tumors has given widely differing ratios. This diversity of results requires explanation and raises questions concerning the validity of estimates as to the number of histocompatibility loci. The experiments cited have also shown curious differences in the survival of skin grafts in segregating generations. Even though many grafts were rejected within the first 2 weeks, others persisted for months before being sloughed. This raises interesting questions about the properties of histocompatibility loci concerned. Moreover, a comparison of the results of this type of experiment with possible theories of graft rejection will show that the assumption of simple dominance is as admissible as the assumption of codominance. Dominance rather than codominance was, in fact, the assumption made in the original theory ( Little, 1941). We must then ask whether the assumption of codominance is necessary. Although the facts presented so far presented can be taken as evidence that the genetic theory of transplantation as set forth above has essential validity, they leave a number of questions unanswered. We shall defer answers to these questions until additional evidence has been assembled.
CONGENIC RESISTANT LINES
Grafts made to F2 and backcross generations provide evidence for the existence of multiple histocompatibility loci, but do not establish the individual identity of the loci. They do not permit any separation of the loci one from another. Such separation has been accomplished by two methods, the first primarily serological, the second primarily genetic. Even though historically the serological method was the first to be employed, the one we shall examine first is the genetic.
Method of production
Coisogenic and congenic strains have already been defined. Congenic strains in which the significant difference is at a single histocompatibility locus can be produced by an appropriate series of crosses, with implantation of tumor to select resistant animals in every second or third generation. Such strains may be called congenic resistant or CR strains, because they resist grafts from their congenic partner. The theory of the production of such lines is discussed in Snell ( 1948), Snell and Bunker ( 1965), and Chapter 2. The cross-intercross system which has usually been employed is illustrated in Figure 24-2. The end result of matings made according to this system is a line, A.B, or a group of lines A.B(1), A.B(2), A.B(3), etc., each of which may be presumed to differ from strain A at a single histocompatibility locus. Perfect coisogenicity is never attained (discussion in Chapter 2), but it is possible to attain an approximation thereto adequate for the purposes for which the lines are designed.
Symbols for designating congenic lines
Symbols for designating inbred strains of mice are discussed in Chapter 6. Because congenic strains of mice present certain special problems not covered by the usual nomenclature rules, some further details are given here.
Congenic resistant (CR) strains are usually designated by a symbol consisting of the symbols of the two strains used in the initial cross, separated by a period. The strain to which all subsequent crosses are made, and which therefore provides the congenic partner to the CR line, appears first. Symbols are usually abbreviated, using standard abbreviations where these are available. Thus a line congenic with C57BL/10 in which the gene for resistance came from BALB/c would be called B10.C. Where several lines are derived from the same cross, these are distinguished by appending a number, or a number and letter, in parentheses. We have used the letter M to mean production by the cross-intercross system, N to mean production by the cross-backcross-intercross system, and NX to mean production by backcrossing a gene with a visible dominant effect, and known to be linked with a histocompatibility gene, onto an inbred background. Thus B10.C(41N) and B10.C(47N) are CR lines produced by cross-backcross-intercross matings in which a histocompatibility allele from BALB/c has been introduced onto a C57BL/10ScSn background. Once the foreign allele in the CR line has been identified, an alternative symbol in the form C57BL/10-H-7b (= B10.C(47N)), in accordance with rule 5 of Strain Rules, Chapter 6, becomes permissible.
Analysis of linkage
Once a group of lines on one or more inbred backgrounds is established, the problem remains of identifying the histocompatibility locus by which each is distinguished. Two CR lines from the same initial cross may be different, but they may also be identical. The process by which each line is produced merely selects an allele at some foreign histocompatibility locus for introduction onto the chosen background; it provides no foreknowledge of what locus this will be.
Two methods of analyzing CR lines once they are established are available. The first of these, described in this section, is analysis by linkage. If a histocompatibility locus can be shown to occupy a particular position on the linkage map ( Chapter 8), it is of necessity different from a histocompatibility locus occupying any other position on the map.
The first step in analysis by linkage may be essentially a fortuitous one. In several instances, at first entirely by accident but in later studies partly by choice, the parents of congenic lines have differed by genes with visible effects closely linked with histocompatibility genes. Such genes serve as "markers." The introduction of the histocompatibility gene onto the inbred background introduces the marker gene also, and in the later intercross generations the linkage becomes apparent through an association of the marker gene with resistance. This has happened a number of times. An example is given in Table 24-5, which shows a clear association of albinism ( c) and resistance found in the 12th generation (an intercross generation) in the production of CR line B10.C(41N). This line was derived from an initial cross between C57BL/10 and the albino strain BALB/c (or actually C.B6, a congenic partner of BALB/c with the H-2 allele of C57BL/6 substituted for the H-2 allele of BALB/c).
Three histocompatibility loci, H-1, H-3, and H-4 have been identified by this means. H-1 and H-4 are in linkage group I, but, as subsequent studies showed, in different positions in this linkage group, and H-3 is in linkage group V. Histocompatibility-2 in linkage group IX would have been identified by this means had it not already been identified by serological tests and by linkage tests of another type. The pertinent congenic resistant strains are shown in Table 24-6.
Once linkages are identified by a chance association of resistance with a marker gene, it is possible to set up crosses specifically designed to give accurate information on the crossover percentage. This has been done for H-1, H-2, H-3, and H-4. The details will not be described here, but some of the results are summarized in Table 24-7 and Figure 8-3. Further information is given under the discussion of the separate loci.
Analysis by the F1 test
More often than not, congenic resistance lines are, so far as all visible traits are concerned, identical with their congenic partners. It is only the exceptional line that carries a distinguishing marker gene. Some other method than linkage must therefore be used for the analysis of most lines.
An effective alternative is provided by the F1 test ( Figure 24-3). In this test, two CR lines on the same inbred background are crossed, and the F1 hybrid is challenged with a transplant from the inbred partner. If the two lines are identical (as in the case of lines 2 and 3 in the figure), the F1 of necessity reduplicates their genotype, and likewise their resistance. If the two lines are different (as in the case of lines 1 and 2 in the figure), the two genotypes complement each other and produce a susceptible hybrid. A special case arises when the two lines come from different initial crosses (strains 3 and 4 of the figure). Here the two lines may differ from the common partner at the same locus, but by different alleles. The hybrid, H-2b/H-2c will, in this case, usually be resistant to H-2a/H-2a donor tissues, but not always. The alleles may complement each other. At least one case is known where the complementation is complete ( Snell et al., 1953). How often complementation occurs is unknown, but it is likely that, at loci where there are multiple alleles, partial complementation is rather common, but complete complementation rare.
If then, two CR lines on the same genetic background give a susceptible hybrid, it may be inferred with considerable confidence that they differ from this common partner at different loci. If one identifies a locus H-1, the other must identify a distinct locus which can be called H-2. If three lines give susceptible hybrids in all three possible F1's, then three loci are identified. A necessary proviso is that the lines be on a common background, though the method can, in theory, be extended to lines on different backgrounds provided alleles are shared in common.
Table 24-8 shows the application of the F1 test to two lines, B10.C(41N) and B10.C(47N), derived from an initial cross between C57BL/10 and BALB/c. As already mentioned, B10.C(41N) is an albino line, and on the basis of the linkage of resistance with albinism is presumed to differ from C57BL/10 at the H-1 locus. Both lines were crossed with a panel of CR tester stocks of known histocompatibility genotypes. Three of the tester stocks B10.129(5M), B10.LP, and B10.129(21M), are listed in Table 24-6. These stocks test for H-1, H-3, and H-4, respectively. The other stocks used were B10.BY which provides an alternative test for H-1 and B10.D2 which tests for H-2. As expected, from prior evidence derived from linkage, line B10.C(41N) gave resistant hybrids with H-1 tester stocks, but positive hybrids with other stocks. The conclusion that it differs from C57BL/10 at H-1, or, as we may say for convenience, that it is an " H-1 line," is confirmed. Line B10.C(47N), on the contrary, gave susceptible hybrids with all stocks. The inference is that it does not differ from C57BL/10 at H-1, H-2, H-3, or H-4, and therefore must differ from it at some previously unidentified histocompatibility locus. Since Amos et al. ( 1963) have assigned the symbols H-5 and H-6 to loci identified by serological methods (see below), the locus identified by B10.C(47N) may be assigned the symbol H-7. The evidence that H-7 is different from H-5 and H-6 will be discussed later.
ANALYSIS OF HISTOCOMPATIBILITY ALLELES
Multiple allelic systems are common in genetics, and it is not likely that histocompatibility loci present an exception. What methods are available for the analysis of histocompatibility alleles? Five approaches have been used; we shall discuss only the three most useful ones. One of these involves an extension of the F1 test we have just described. We shall examine this one first.
As applied to the identification of alleles, the F1 test employs a CR pair and an unrelated inbred strain, the unknown. The three strains may be represented as follows:
| Tissue donor | Known parent | Unknown parent | ||
| A | A.B | U | ||
| (A) H-1a | (A) H-1b | (U) H-1x |
Relevant information as to the genotype of the three lines is given in the formulae (A) H-1a, (A) H-1b, and (U) H-1x, where H-1 is the locus at which strains A and A.B differ, (A) is the total genotype of strain A exclusive of H-1, (U) the same for strain U (the unknown), H-1a and H-1b, the known H-1 alleles of strain A and A.B respectively, and H-1x, the unknown H-1 allele of strain U. If strains A.B and U are crossed, the formula of the F1 hybrid can then be written (A)/(U) H-1b/H-1x. It is apparent that if H-1x is, in fact, H-1a, this hybrid will be susceptible to strain A transplants since every gene found in strain A can also be found in the F1. When H-1x is not H-1a the situation is more complex. Usually the transplant will be rejected, but there is also the possibility that H-1b and H-1x will show a complementary relationship to H-1a. Thus if each allele determines antigenic specificities as follows:
| (A) H-1a | (A) H-1b | (U) H-1x | ||
| 1,2,3 | 1,-2,-3 | -1,2,3 |
The F1 test can also be reversed, using A.B as the tissue donor and A as the known parent. The allele tested for is then H-1b. It is also possible to treat the tissue donor as the unknown and the second parent as the known.
An example of the typing of 10 inbred strains (the unknowns) by the F1 test is shown in Table 24-9. The tissue donor is strain C57BL/10 and then known parent CR strain B10.C(47N). Since the donor and the known parent differ at H-7, with the allele of the donor being H-7a, this is a test for H-7a. Of the 10 hybrid combinations, seven proved susceptible, all mice succumbing to the tumor, and three resistant. The parents of the resistant F1's were strains A, BALB/c, and C3H. Since the gene for resistance in strain B10.C(47N) came from strain BALB/c there would have been an inconsistency had BALB/c not produced a resistant F1. Presumably the H-7 alleles in BALB/c and B10.C(47N) are the same, and both may be designated H-7b. All that has been proved formally, however, is that strains A, BALB/c and C3H do not carry H-7a. A considerable presumption is also established that the other seven strains are H-7a, though there is a possibility that one or more of them carries and H-7 allele different from both H-7a and H-7b, but sufficiently like H-7a so that any missing components of H-7a are supplied by H-7b.
Table 24-10 gives information on the distribution, within a number of inbred and CR strains, of known alleles of H-1, H-3, and H-7. (For comparable information on H-2 see Table 24-13.) It should be remembered that evidence that a particular strain does not carry a certain allele is more conclusive than evidence that it does carry it. The information in Table 24-10 on alloantigenic specificities will be discussed later. An interesting sidelight provided by this table is the tendency of lines known to be related to have similar histocompatibility genotypes (e.g., C57BL/10, C57BR/cd, C57L, and C58; A and BALB/c; DBA/1 and DBA/2).
The primary value of the F1 test in studies of allelic systems is in typing a variety of strains for already identified alleles. By showing that a given strain lacks the alleles of both members of a congenic pair, a third allele can be proven, but beyond this the method cannot go. Other methods are necessary for establishing longer series of alleles.
A useful method may be available when multiple congenic resistant lines on a common background have been produced by more than one initial cross. Sometimes these lines will differ from the background line at the same locus but by different alleles. This happened with H-1, partly by design in this instance since albinism could be used as a marker. The relevant lines are B10.BY, B10.129(5M), and B10.C(41N). Evidence from the F1 test and from linkage of resistance with the albino locus prove that these all differ from C57BL/10 and H-1. One method of demonstrating different alleles in such lines is to exchange skin grafts between them. Strains with different alleles should be histoincompatible. This method is straightforward but requires a high degree of coisogenicity in the lines if the results are to be trustworthy. This method has not been used extensively to date but undoubtedly will be used more in the future.
A third method of analyzing alleles uses alloantisera and is essentially identical with the method used for the study of blood groups in man. This method is described in connection with the discussion of H-2.
Information on two other methods of studying histocompatibility alleles not described here will be found in Snell et al. ( 1953) and Snell et al. ( 1957).
Evidence concerning the multiplicity of alleles is summarized in Tables 24-10 and 24-13 and discussed in connection with the descriptions of the separate loci.
In concluding this section on histocompatibility alleles, it should be reemphasized that, whenever the method used, evidence of dissimilarity of alleles is more conclusive than evidence of identity.
Once pairs of congenic resistant lines with single histocompatibility differences were available, it became possible to characterize individual histocompatibility loci in a way that had been quite impossible when all information was based on comparisons of inbred strains with multiple differences or the segregating generations derived therefrom. Not surprisingly, the different loci showed marked individuality.
DIFFERENCES IN "STRENGTH" OF HISTOCOMPATIBILITY LOCI
Once pairs of congenic resistant lines with single histocompatibility differences were available, it became possible to characterize individual histocompatibility loci in a way that had been quite impossible when all information was based on comparisons of inbred strains with multiple differences or the segregating generations derived therefrom. Not surprisingly, the different loci showed marked individuality.
One of the first differences that became apparent was in the magnitude of the barriers which different loci oppose to transplants. Some of the barriers are easily transgressed, others are transgressed with great difficulty. This has been referred to as a difference in the "strength" of the loci ( Counce et al., 1956). This is not an altogether happy expression, but the usage is now well established, and in the absence of any better alternative we shall follow it here.
Some results with tumor transplants made from strain C57BL/10 to 15 lines congenic with C57BL/10 and with skin grafts exchanged in both directions between C57BL/10 and the same lines are shown in Table 24-11. It should be noted that all tumors were transplanted to hosts immunized three times with C57BL/10 tissue and that the tumors were selected for their specificity. Without prior immunization or with other tumors more deaths would have resulted. Mice receiving skin grafts were unimmunized.
It will be seen that there is a great difference in the behavior of the transplants in the different lines. In several lines all animals survived the tumor transplants; in other lines deaths ranged up to 78 per cent. There were similar differences with skin grafts. Grafts from C57BL/10 to strain B10.D2 showed a median survival of 9 days, grafts from C57BL/10 to B10.129(21M) a median survival of 127 days. In the weaker combinations there was great variability in the interval to rejection. In these combinations also chronic rejections, characterized by apparently incipient rejection followed by partial recovery, were common. In general there was a close correlation between the percentage of deaths from tumor grafts and the length of survival of skin grafts.
The data from skin grafts bear on the question of whether histocompatibility loci show dominance or codominance. In every instance where skin grafts were made reciprocally within a strain pair they were rejected in both directions. There was a great difference in the two directions in some instances, but never 100 per cent survival in either direction. Thus with the strain pair B10.129(5M) and C57BL/10, most of the grafts survived beyond 100 days when strain C57BL/10 was the host, though only 28 days in the reciprocal direction. It follows that none of the alleles tested can be regarded as determining the absence of an antigen (though some may approximate this). It also follows, since grafts from any pair of strains grow in the F1 (one exception will be discussed later), that the alloantigens determined by each strain are expressed in the F1. This constitutes codominance by definition.
Other aspects of Table 24-11 will be considered when the separate loci are discussed.
THE H-1 LOCUS
The H-1 locus is defined by a group of congenic resistant strain pairs (Tables 24-6 and 24-11) which have been shown to share a common difference and by its linkage with albinism. At least four alleles have been identified ( Table 24-10) and it seems likely that there are others as yet undetected. Skin allograft rejection in H-1 CR pairs ranges from a median of 15 days for grafts made from C57BL/10 to B10.BY to more than 100 days for grafts made from B10.129(5M) to C57BL/10 ( Table 24-11). As already noted the last named pair shows a marked reciprocal difference.
Ovarian grafts exchanged between H-1 pairs survive much longer than skin grafts; probably they survive permanently in at least some instances ( Snell and Stevens, 1961). This is probably a peculiarity of the tissue, not of the locus, since long survival of ovarian grafts across other non- H-2 barriers has been noted ( Linder, 1961). The H-1 locus is a blood group locus as well as a histocompatibility locus. Appropriate antisera will distinguish between the erythrocytes of the congenic partners C57BL/10 and B10.129(5M) (Stimpfling, unpublished data; Snell and Graff, unpublished data).
THE H-2 LOCUS
History
It is generally accepted that of the dozen or more histocompatibility systems known in the mouse, the histocompatibility-2 or H-2 system of linkage group IX is essentially unique in both its complexity and in the immunogenic complexity and potency of its end product. The term H-2 is derived from antigen II, a cellular antigen initially identified by Peter A. Gorer with the aid of a rabbit antimouse serum. Gorer also demonstrated that the rejection of a tumor allograft resulted in the formation of humoral antibodies specific for antigen II present on the erythrocytes of the tumor-inducing strain. Further, it was shown by tumor grafting and blood typing mice of the F2 and the appropriate backcross generations that antigen II was probably determined by a dominant gene and that compatibility of the tumor and host with respect to antigen II was a necessary but not entirely sufficient condition for the progressive growth of the graft (Gorer, 1937, 1938, 1942). The observations of Gorer provided direct evidence of an immune reaction to allotransplants and demonstrated the existence of at least one antigen system shared by a tumor and the normal cells of the strain to which the tumor was indigenous.
Genetic tests made by Gorer et al. ( 1948) showed that there is a linkage between H-2 and the gene Fu located in linkage group IX, and further tests by Allen ( 1955a, 1955b) served to locate H-2 with respect to other genes in this linkage group ( Table 24-7 and Figure 24-4).
The linkage of H-2 with Fu and T has been used to detect alleles at the H-2 locus. The method will not be discussed here as it has been fully described elsewhere ( Snell et al., 1953). Originally three variants were identified and assigned the symbols H-2, H-2d, and h-2. Strain A was classified as H-2, strain DBA as H-2d, and strain C57BL as h-2. The symbol h-2 with a small letter was used because in the early studies no alloantigenic activity was detected in the red cells of mice with this allele. Several other strains were tentatively put in this category. Subsequently all alleles were found to determine effective alloantigens and show codominance. The symbol h-2 was therefore dropped and replaced in all cases with H-2, with a superscript small letter added to indicate the allele. The number of identified alleles was also greatly increased. As of 1965, 20 alleles are positively known. However, tests of a randombred strain showed it to be segregating for a number of alleles as yet unidentified ( Rubinstein and Ferrebee, 1964). Undoubtedly, the ultimate number of alleles will greatly exceed 20.
Early studies, both serological and genetic ( Gorer, 1938; Gorer et al., 1948; Snell et al., 1953), demonstrated that the H-2 alloantigen(s) is characterized by multiple components or specificities. Thus Gorer showed by absorbing a C57BL anti-A serum with CBA cells that the serum contained two specificities. Probably both specificities were determined by H-2, though this was not proved at the time. Subsequently it was shown in transplantation experiments that all F1 hybrids of the genotype H-2d/ H-2k, whatever the source of these two alleles, were susceptible to transplantable strain A tumors of the genotype H-2a/ H-2a. This led to the suggestion that the alleles H-2d, H-2k, and H-2a determined, respectively, the "components" D, K, and DK ( Snell et al., 1953). D was probably identical with Gorer's original antigen II. The presence of K in strains typed genetically as H-2k or H-2a was later demonstrated by serological methods also.
Serological studies
Much of our information concerning the H-2 locus has been derived from serological studies. One of the surprises of histocompatibility in the mouse is the ease with which alloantibodies reactive with H-2 specificities are produced and demonstrated, and the relative difficulty of producing, or at least of demonstrating, alloantibodies reactive with the specificities determined by other loci. Red-cell agglutination has been by all odds the most useful serological technique for studying H-2. A limited use has also been made of the cytotoxic test which employs lymphocytes as the target cells. These tests have shown that the alloantigen determined by H-2 is remarkably complex. Because they reveal individual specificities, or at least can reveal such specificities when appropriate antisera are used, they nicely complement the methods of tissue transplantation which respond to groups of specificities more nearly representative of the totality of a given allele. While Gorer first demonstrated the H-2 antigen by use of rabbit antisera, all studies are now carried out with alloantisera produced in one strain of mice by the injection or transplantation of the tissue of another strain.
It was observed early in the investigation of H-2 alloantigens that strong hemagglutination reactions in saline media could be obtained routinely only with cells of certain strains while the results with cells from other strains often gave irregular reaction. Further, some sera of high titer became inactive following storage for a few hours while others retained their activity after many weeks of storage. Subsequently, it was found that some apparently inactive antisera agglutinated erythrocytes if the cells were suspended in human serum. A further improvement in the test consisted of diluting antisera in an appropriate dextran solution and suspending red blood cells in normal human serum previously absorbed with mouse tissue ( Gorer and Mikulska, 1954). A modification of this test was described in which cells are suspended in saline and antibody diluted in polyvinylpyrrolidone or PVP ( Stimpfling, 1961). The use of developing or conglutinating agents such as dextran and PVP in mouse hemagglutination tests in necessitated by what appears to be the "incomplete" character of mouse alloantibodies.
Besides the hemagglutination test, several other serological techniques have been used to study one or another aspect of histocompatibility phenomena. Papers describing leukocyte agglutination tests, cytotoxic tests, hemolytic tests, the fluorescent antibody technique, and various other methods relevant to the serological study of histocompatibility genes and antigens will be found in the Bibliography of Techniques at the end of this book.
A serological technique useful with mice is absorption in vivo. Animals of the appropriate genotype are injected intraperitoneally with 0.2 to 0.3 ml of antiserum and bled 2 or more hours later. The method is much simpler than absorption in vitro with liver or red cells, and with a good antiserum the lowered titer presents no difficulties.
The immunization of mice with allogeneic tissues or purified tissue derivatives usually results in the formation of antibodies detectable by one or more of the above techniques. Usually these antibodies consist of a mixture of specificities. Since much of the value of the serological method lies in its ability to reveal single antigenic components or specificities, it is important to examine the method by which individual specificities are detected.
Gorer and Mikulska ( 1959) have shown that there is a simple mathematical relationship between the number of genetically different inbred strains available for testing and the maximum number of antigenic specificities, other than specificities related to sex, that can be detected. The requirements for the detection of different specificities are: (1) that with respect to available strains they be present in at least one and absent in at least one, and (2) that with respect to the available strains their distributions show at least one difference. With two strains not more than two specificities are demonstrable. The situation for four strains is illustrated in Table 24-12. It will be seen that in this situation the theoretical total of demonstrable specificities is 14. More generally, the maximum number of specificities that can be revealed with n inbred strains is the sum of the number of combinations of (n - 1) in n, plus the number of combinations of (n - 2) in n, plus the number of combinations of (n - 3) in n, etc., or
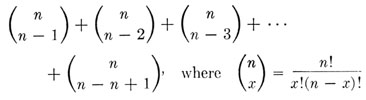
The terms of this formula developed by Gorer and Mikulska, as it so happens, are the coefficients of the binomial expansion, exclusive of the first and last which are both 1. Since the sum of these coefficients is 2n, the above formula is equivalent to 2n. If we apply these two formulas to the situation illustrated in Table 24-12, where n = 4, we find that the maximum number of demonstrable specificities equals 4!/(3!1!) + 4!/(2!2!) +4!/(1!3!) = 24 - 2 =14. It will be seen that as n increases the number of detectable specificities increases rapidly. With 10 strains it becomes 1,022. It should be emphasized that this is the theoretical maximum. Because the distribution of specificities in any group of strains will virtually never conform to the optimum pattern, and because antibodies are produced less easily to some specificities than to others, the actual values will almost always fall below that theoretically obtainable. It should also be emphasized that the mere detection of specificities tells nothing about the locus by which they are determined. Actually most of the easily demonstrable specificities in the mouse are directed against H-2, but added sources of information are necessary if specificities are to be related positively to a given locus. The easiest of all methods for accomplishing this is to use as donor and recipient a congenic strain pair with a single known histocompatibility difference. Such pairs have been used extensively to produce antisera whose specificities are limited to H-2.
Most antisera prepared against allogeneic tissues contain multiple specificities. The problem of producing monospecific antisera can be illustrated by further recourse to Table 24-12.
An antiserum prepared in strain A against strain B may contain antibodies specific for components 1, 6, 8, and 12. The specificity of this antiserum can be increased by appropriate absorption, either in vivo or in vitro, with the tissues of a third, crossreactive strain. For instance, absorption of strain A anti strain B serum with tissues from strain C will selectively remove antibodies specific for components 1 and 6, but leave behind antibodies specific for components 8 and 12. A further refinement in the preparation of serotyping reagents can be attained by producing antisera in hybrids. If, for example, an antiserum is made in hybrids between strain A and strain C against tissues of mice from strain B, the antibodies will be specific for components 8 and 12. An anti-12 serum can be obtained by absorption in vitro or in vivo with strain D tissue. Actually this antiserum may still not be monospecific, but, in addition to its reactivity with 12, may be capable of reaction with one or more specificities not indicated in Table 24-12 and capable of absorption only by recourse to additional strains.
By use of procedures outlined above, 30 or more serologically distinct specificities have been detected on mouse red cells. The majority of these are determined by the H-2 locus.
The specificities which characterize the H-2 alloantigen were originally designated by capital letters. Ultimately, however, the number of reported specificities outran the letters of the alphabet, and it became necessary to assign compound symbols such as A1 and B1 ( Stimpfling and Pizarro, 1961). Such symbols are awkward and confusing. We are therefore adopting a numerical system of nomenclature proposed later ( Snell et al., 1964). This parallels and is largely based on a similar system proposed fro the Rh blood group of man ( Rosenfield et al., 1962). Each specificity is designated by an Arabic numeral, its absence by a minus sign (-), or, where the minus sign alone would not be clear, by the appropriate numeral preceded by a minus sign, e.g., -7, -8, etc.
The H-2 chart
Much of our knowledge of the < I>H-2 locus can be summarized in the form of a chart which lists (1) the known H-2 alleles, (2) the antigenic specificities which have been found associated with the different alleles, and (3) the inbred strains carrying each allele. Such a chart is presented in Table 24-13.
It will be seen in the table that there are several gaps in the listing of specificities. Numbers 15, 18, 20, 21, 23, 24, and 26 are vacant. Most of these gaps are due to current uncertainty about the specificities to which these numbers (or the corresponding letters) were assigned. Two of the missing specificities, 18 (R), identified by an RIII anti-C3H antiserum, and 26 (Z), identified by a C3H anti-RIII antiserum, are determined by a gene or genes believed to be in linkage group IX but distinct from H-2 ( Hoecker and Pizarro, 1962). They, therefore, do not belong in the H-2 table. Shreffler (1965, personal communication) believes that specificities 6 and 28 may be the same. Contrary to previous reports, he finds that strain AKR.M reacts with (will absorb) an anti-6, whereas strain P does not react with it.
The H-2 table provides a convenient summary of available knowledge concerning H-2, but it fails to reveal a number of known complexities of H-2 immunogenetics. These complexities constitute pitfalls concerning which anyone contemplating experimental use of H-2 should be forewarned. Not all of these can be described here, but a few salient facts must be mentioned.
The alloantigenic specificities determined by H-2 differ in their "strength" as indicated by the ease with which alloantibodies are induced, by the titer of the resulting antiserum, and by the ease and consistency with which agglutination is obtained when presumably active sera are titered against reactive red cells. While there is no sharp division of the specificities into "strong" and "weak" groups, specificities, 1, 2, 3, 4, 5, 9, 11, 16, 17, and 19 tend to be easily demonstrable. In donor-recipient combinations differing at both strong and weak specificities, antibodies to the latter may appear late or not at all. Sometimes the same specificity behaves differently on different genetic backgrounds. Often specificities which cannot be demonstrated by agglutination can be demonstrated because antibodies are removed by absorption.
Curiously, two H-2 specificities, 32 and 33, have so far been demonstrated only by the cytotoxic test ( Gorer, 1959). They are, therefore, present on lymphocytes, but are absent from, or at least difficult to demonstrate on, erythrocytes.
There are probably minor variants of some H-2 alleles. By their very nature there is more uncertainty about these variants than about the major differences. None are included in the table. One of them, H-2d', has been detected serologically ( Gorer, 1956) as well by the methods of tissue transplantation ( Snell et al., 1953). This variant occurs in strains YBL and YBR. The distinguishing specificity, called D', is also omitted from the table. There are probably minor variants of H-2k, but how these are distributed among the different strains typed as H-2k is undetermined ( Snell et al., 1953).
It has already been noted that there is an unusual relationship between the alleles, H-2d, H-2k, and H-2a, in that the hybrid H-2d/ H-2k, whatever may be the source from which these alleles are derived, is susceptible to strain A ( H-2a/ H-2a) transplantable tumors. This complementarity of H-2d and H-2k is also revealed in the specificities listed in Table 24-13. Allele H-2a has no specificities not present in alleles H-2d and H-2k, and, with the exception of specificities 31 and 32 which occur in H-2d and H-2k, respectively, but not in H-2a, has all he specificities present in H-2d and H-2k. If we assume that 31 and 32 are not represented in H-2a by some alternative specificity but rather by a true absence of antigenicity, all the requirements of the compatibility relationships of these strains are met. Of course hybrids carry incompatibilities at other loci, but these are overridden by the rather virulent transplantable tumors which have been used in these tests.
It has been suggested that the unusual relationship of these three alleles can be explained by the assumption that H-2a was derived from a crossover within the H-2 locus occurring in a mouse heterozygous for H-2d and H-2k ( Gorer, 1959).
One of the interesting questions about H-2 specificities is whether any of them are "allelic." The concept of allelism was originated in connection with alternative or mutually exclusive genes occupying the same locus. It could reasonably be applied to alloantigenic specificities if it could be shown that these sometimes occupy the same site on the antigen molecule and are therefore mutually exclusive. The extension of the term would gain added justification if proof were also forthcoming that mutually exclusive specificities were determined by mutually exclusive or allelic subunits of a complex gene. No firm information of this sort is available concerning the specificities determined by H-2. However, it will be seen from the table that some specificities do appear to show a mutually exclusive distribution. Thus specificities 5 and 31 have never been found in combination 2. Partly because of this 5 and 31 were originally assigned the symbols E and Ed, implying an allelic relationship. While allelism of specificities may ultimately be proved, the evidence now available is of a very uncertain nature, and we have followed Snell et al. ( 1964) in treating all specificities as nonallelic.
A serologically detected serum variant determined by a locus designated Ss, although without known effects on histocompatibility, is of interest in connection with H-2 because of its linkage relations. Two alleles are known. Ssh determines a high level of a specific serum protein, Ssl a low level. Numerous inbred strains have been typed for Ss. All strains which are H-2k, and AKR.M, the only known H-2m strain, are Ssl; all other strains as Ssh ( Shreffler and Owen, 1963; Shreffler, 1964, 1965). The unusual linkage relations of H-2 and Ss are described below.
Crossing over within H-2
Crossing over within the H-2 locus was first observed by Allen ( 1955b) and by Amos et al. ( 1955b). Allen, using progeny tests and tumor transplants to type animals from a cross H-2a + T/H-2f Fu + x H-2b + +/ H-2b + +, found one proven and one doubtful crossover between "components" 4 and 11 in 284 mice. Amos et al., using red cell agglutination, absorption in vivo, and progeny testing to type the animals from a cross H-2b/ H-2a x H-2b/ H-2b, found one crossover between specificities 2 and 5 in 32 mice. Subsequently the initial report of Amos et al. was extended by additional publications ( Gorer, 1959; Gorer and Mikulska, 1959; Amos, 1962; see also Table 24-13 for the complete serotype information on the three crossovers reported in these studies), and three other crossover studies were carried out ( Pizarro et al., 1961; Shreffler, 1964, 1965; Stimpfling and Richardson, 1965).
Fourteen crossover alleles obtained in these studies, each of which has been typed for five or more specificities, are shown in Table 24-14. Four of the crossovers were also typed for Ss ( Shreffler, 1965). It will be seen that these crossovers serve to identify five regions within the H-2 locus or "complex," and that they place Ss within H-2. The regions and the specificities assigned to each are D (2,4,13), C (3), V (22), E (5), and K (11,19,31). Ss lies between E and K. Information as to specificities not shown in the table also locates 1 and 8 in C, V, or E. So far as is known, the Ss protein is neither antigenically similar to nor structurally associated with the H-2 antigen. If this lack of relationship is confirmed by further studies, it will be necessary to regard the K region of H-2 as a separate locus.
It was suggested above that allele H-2a was derived by crossing over between alleles H-2d and H-2k. If this is indeed the origin of H-2a, strains which are H-2a should be Ssl whereas actually they are Ssh. The meaning of this discrepancy is not clear at the present time.
The percentage of crossing over between three of the regions, based on the study of Stimpfling and Richardson ( 1965), is shown in Table 24-15. The values range from 0.55 for the D-K interval to 0.05 for the C-K interval, both in heterozygous females. In accordance with results usually obtained in the mouse, crossing over, for two of the intervals, is substantially more frequent in heterozygous females than in heterozygous males, but the differences are not statistically significant. Shreffler ( 1965) has reported similar values. Somewhat higher values have been reported by other investigators but these were based on many fewer animals.
The crossover obtained by Allen ( 1955b) indicated that the order of the H-2 regions in relation to the genes Fu and T of linkage group IX is D K Fu T. This order has subsequently been confirmed ( Stimpfling and Richardson, 1965; Shreffler, 1965). Information in regard to H-2 and its relation to other identified loci in linkage group IX is shown in Figure 24-4. Other factors shown in the figure in close association with H-2 will be discussed later.
Transplantable tumors induced in hybrids between two inbred lines usually fail to grow in either parental strain. If the H-2 antigens of one parent are lost from the hybrid tumor, the tumor is then able to grow in the other parental strain. This provides the basis for an assay system used by several investigators to select variants lacking all or some part of one of the parental H-2 complexes ( Mitchison, 1956; Hellström, 1961; Dhaliwal, 1964; Klein and Klein, 1964). On the basis of serological and transplantation criteria, it has been shown with several types of tumors, including sarcomas, carcinomas, and leukemias, that the loss of H-2 alloantigens was persistent, irreversible, and specific. Hellström ( 1961) tested 12 variants of two (A x A.SW)F1 lymphomas and found that 11 had lost specificity 11 and one had lost both specificities 4 and 11. The loss of 4 without 11 has not been observed. The loss of alloantigens from hybrid tumors is compatible with the interpretation that the variants arose by mitotic crossing over. However, other genetic mechanisms cannot be excluded. In any case, these observations are consistent with the existence of functionally independent regions of the H-2 locus. They also suggest, since 11 can be lost without 4 but not 4 without 11, that the kinetochore or spindle-fiber attachment is adjacent to the D (4) rather than the K (11) region of H-2. This is the basis for the position assigned to the kinetochore in Figure 24-2.
The H-2 system has intrinsic interest, apart from the role that it plays in tissue transplantation, as a model for the study of complex genes in mammals. Studies on the serological and genetic characteristics of H-2 may provide some insight into the immunogenetic properties of similar types of blood-group systems in other vertebrates.
Chemistry of the H-2 alloantigen
Because of the unique role which H-2 plays in graft rejection, most efforts to isolate an alloantigen from mouse tissues have centered on the products of this locus. Several methods of extraction have been employed to obtain immunologically active, cell-free preparations from both normal and tumor tissues ( Castermans and Oth, 1959; Herzenberg and Herzenberg, 1961; Brent et al., 1961; Manson et al., 1963; Kandutsch and Stimpfling, 1963, 1965). Assays of the various preparations for biological activity have included tests of the ability to induce accelerated rejection of skin allografts, elicit the formation of hemagglutinins, absorb hemagglutinins from appropriate antisera, and abrogate resistance to tumor allografts (immunological enhancement).
A variety of tissues has been used as a source of alloantigens, including spleen, lymph nodes, liver, and transplantable tumors. Irrespective of the source of starting material, procedure of extraction, or assay system used, the available information indicates that the major part of H-2 activity is associated with the membranous structures of cells.
The earliest studies on the properties of transplantation antigens were carried out by Snell ( 1952). At that time the relation of the immunologically active fractions to H-2 was unknown. Kandutsch undertook a study of the chemical properties of the enhancing or H-2 substance in 1954 and has succeeded in preparing a soluble alloantigenic lipoprotein in a relatively homogeneous form, according to electrophoretic and ultracentrifugal criteria ( Kandutsch and Reinert-Wenke, 1957; Kandutsch and Stimpfling, 1963, 1965). The preparation described by Kandutsch was obtained from a particulate fraction of a strain A tumor by extraction with the detergent, Triton. In the presence of Triton, its sedimentation coefficient suggested a relatively low molecular weight. In the absence of Triton and at a pH in the region of neutrality, the material was almost completely insoluble in water. Digestion with phospholipase A extracted from snake venom rendered the Triton-soluble lipoprotein soluble in water with no gross change in lipid or amino acid composition. Alloantigenic activity of both the Triton-soluble lipoprotein and the product obtained by venom-digestion was demonstrated by the hemagglutinin production, hemagglutination-inhibition, accelerated skin graft rejection, and enhancement tests. The preparations were shown to have specific activity corresponding to the H-2 specificities 4, 8, and 11. The activity appeared to be independent of the lipid content of different preparations, suggesting that it resided largely or entirely in the protein component.
The investigation of the chemical properties of histocompatibility antigens is of considerable interest, not only because of their role in tissue transplantation but also because some or all of these antigens are components of cell membranes and probably contribute in varying degrees to maintaining the structural and functional integrity of living cells.
THE H-3 COMPLEX
The H-3 locus was originally defined by the congenic lines C57BL/10 and B10.LP, nonagouti ( a) and white-bellied agouti ( Aw), respectively, in their coat colors, and by the association, demonstrated by crosses of these lines, of white-bellied agouti with resistance. Crossing over between the a locus and H-3 was estimated at about 10 per cent ( Snell, 1958b).
This rather close linkage between a dominant gene affecting coat color and a gene for resistance provided a favorable situation for further analysis of the H-3 locus. +, Aw, or at from various sources was introduced onto a C57BL/10 background, and the resulting lines subjected to appropriate tests. In most instances the resulting lines resisted transplants from C57BL/10, and linkage tests showed that the resistance was due to a gene or genes associated with the introduced "marker" gene. In two instances, the introduced marker was lost by crossing over, but resistance remained and was shown to be due to a gene close to the a locus. Two of the resulting lines B10.129 (13M), a nonagouti line, and B10.129(14M) a white-bellied agouti line, came from the same initial cross. Both these lines resist transplants of C57BL/10 tissues, and the resistance has been shown to be due to a gene in linkage group V, but the F1 hybrid between them is susceptible. Both give resistant F1 hybrids when crossed to strain B10.LP. Somewhat similar results have been obtained with other lines ( Snell and Bunker, 1964; Snell, unpublished data).
Further tests will be necessary before the meaning of these results is finally known, but the probable interpretation is that there are two rather closely linked histocompatibility loci in linkage group V. Line B10.129(14M) differs from C57BL/10 at the H locus closer to a, lines B10.129(13M) and B10.LP- a at the locus farther from a. Lines B10.LP differs from C57BL/10 at both loci.
By the use of strain B10.UW which carries the linked genes at, un, and we on a C57BL/10 background, one locus of the H-3 complex has now been located definitely close to we, and on the side of we away from a ( Snell and Bunker, 1964; see also the linkage map in Figure 8-3). This locus will retain the symbol H-3. No symbol has been assigned to the other postulated locus.
Until the possible multiplicity of H loci in linkage group V is resolved, the question of possible multiple alleles at H-3 cannot be tested. However, a group of standard inbred stocks has been typed for H-3a, using as the "known parent" a line which should provide a test for H-3 only, with the results shown in Table 24-10. The only standard inbred strains sharing H-3a with C57BL/10 are C57L and C57BR/cd; all other lines tested carry some other allele.
Skin grafts made from C57BL/10 to either B10.LP or B10.LP- a survive about 3 weeks, grafts made in the opposite direction about 5 weeks ( Table 24-11). The similarity of results in the two combinations is interesting since, as indicated above, strain B10.LP apparently has an H difference from C57BL/10 not shared with B10.LP-a as well as one shared with this strain. Curiously, also, skin grafts exchanged between B10.LP and B10.LP- a survive for very long periods ( Berrian and McKhann, 1960b; Graff and Snell, unpublished data) despite the proven difference between the strains.
Attempts to find red-cell agglutinins associated with H-3 have been unsuccessful (Amos, Stimpfling, and Snell, unpublished data).
THE H-4 LOCUS
H-4 is identified by the congenic pair C57BL/10 and B10.129(21M) and by its linkage with pink-eye ( p). No proven crossovers between H-4 and p have occurred ( Snell and Stevens, 1961). Skin grafts exchanged between members of this strain pair showed a median survival time of 127 days when B10.129(21M) was the donor. This is one of the most striking differences between reciprocal grafts that has yet been observed ( Table 24-11).
H-5 has been established by Amos et al. ( 1963) by serological techniques. It is defined by antisera, produced in strain C57BL against transplantable lymphoma 6C3HED, which are reactive with the red cells of certain inbred strains but not of others. Reactivity was tested either by red-cell agglutination or by the usually more reliable method of of removal of agglutinating activity by absorption. When absorption was used, the preferred red cells for testing residual activity were those of strain 129. When red cells of certain other strains were used, they apparently reacted not only with H-5 antibodies but with antibodies of other specificities which these antisera contained, thereby confusing the results.
Table 24-10 summarizes some of the results. In this table the symbol + means that the red cells of the indicated strain were reactive, the symbol - that they were nonreactive. Amos et al. regard reactivity as indicative of the presence of a specific allele, H-5a, but this conclusion rests on the assumption at H-5 is very unlike H-2, determining only one specificity per allele rather than the multiplicity of specificities determined by H-2. It is not clear that this assumption is justified by the evidence so far available. This is the reason for using + and - in Table 24-10 to summarize the evidence concerning H-5 and H-6 which have been established by serological methods, rather than by the letters, indicative of alleles, which are used to summarize the evidence from loci studied by transplantation methods.
It will be seen from the Table that the H-5 antisera reacted with cells of strain 129, but did not react with the cells of strains C57BL, B10.129(5M), or B10.129(21M). Since the H-1 and H-4 alleles, respectively, of the two last named strains came from strain 129, this distribution of reactivities proves the nonidentity of H-5 with H-1 or H-4. Were the antisera in question reactive, for example, with H-1, B10.129 (5M) would have to react like 129. Nonidentity of H-5 with H-3 was proven by showing absence of linkage between H-5 specificity and the a locus. Similarly nonidentity with H-2 was proven by independent segregation of H-5 and H-2 specificities. In these linkage tests, the H-5 specificity segregated in approximately the ratios expected of a unit mendelian factor. These data adequately establish H-5 as a new locus.
It should be noted, however, that the tests of H-5 characterize it as a blood group locus, not necessarily as a histocompatibility locus. Amos et al. believe that it is a histocompatibility locus because they have found the H-5 alloantigen on numerous tissues in addition to the erythrocytes. This is strong presumptive evidence, but final proof must probably await the establishment of congenic lines with an H-5 difference and tests with actual transplants.
Evidence for H-6 is of essentially the same nature as evidence for H-5, though proof that it is distinct from previously identified loci is somewhat less satisfactory ( Amos et al., 1963). The preferred sera for tests for H-6 specificity are made in strains C3H/St against the C3H/He ascites sarcoma MC2M. Evidence that H-6 is not H-2 or H-3 comes from linkage data and should be conclusive. Evidence with respect to H-1 and H-4 comes from the data summarized in Table 24-10 on the strain distribution of alleles and specificities. To evaluate this with respect to H-6, we need to take stock of just what these data can tell us.
The serological methods used to study H-5 and H-6 reveal specificities, not alleles. Specificities and alleles would, in effect, be the same thing if single alleles determined single specificities. Whether this event occurs and, if so, with what frequency, we do not know. Certainly it is very far from being the case at H-2. To further complicate matters, it is difficult to be sure that antisera are monospecific. An antiserum with two specificities may react with three different alleles. Thus anti-(1,2) will react with (1,2), (1,-2), and (-1,2). The F1 transplantation test also does not type for alleles, but rather for a combination of specificities. Evidence from the F1 test that two alleles are different should be conclusive. Evidence that they are the same is not conclusive, but proves that at least one and sometimes a considerable number of specificities are shared in common. With these two distinct typing systems, each with its own deficiencies, how can information be interconverted between them?
Unfortunately the answer is that this can usually only be done with assurance under rather special circumstances. The key situation is as follows. If two strains are known positively to carry identical alleles at a certain locus, and if these strains react differently with an antiserum, then the antiserum cannot be testing for the locus in question. An examination of the data in Table 24-10 will show that H-5 probably does meet this requirement with respect to H-1 and H-4, but that H-6 does not. In fact the " H-6" specificity could very well be one of the specificities determined by H-1. At the time that Amos et al. published their report there was some evidence to the contrary. C3H/He which is plus with respect to H-6, and DBA/1 and DBA/2 which are minus, were all originally reported as carrying allele H-1a ( Snell and Stevens, 1961). As noted above, however, evidence that alleles are the same is less conclusive than evidence that they are different, and subsequent tests have classified DBA/2 as H-1d (Snell and Graff, unpublished). DBA/1 has not yet been retyped. The distinctness of H-1 and H-6 is therefore uncertain. As Amos et al. point out, it is not even entirely certain that the H-5 and H-6 specificities are not themselves the product of one locus, though differences in the tissue distribution of corresponding alloantigens (see below) make this unlikely.
The evidence that H-6 is a histocompatibility locus as well as a blood-group locus is the same as in the case of H-5.
OTHER BLOOD GROUP LOCI
Loci H-5 and H-6 are blood group loci believed to be concerned also with histocompatibility because their antigens occur in tissues other than blood. Evidence has been presented which may identify four other blood group loci. The degree of overlap between blood group and histocompatibility loci is very uncertain, but we describe these additional loci briefly because a relation to histocompatibility may ultimately be demonstrated. It is already known that the antigens determined by some of them are in tumors or in normal tissues other than blood.
Spencer et al. ( 1964) have reported two alloantigenic specificities, Kappa and Iota, identified by alloantisera against certain strains of mouse cells grown in vitro. The antibodies were demonstrated by red cell agglutination, but the cells that induced them were not of hematogenous origin, so the antigens seem not to be confined to blood. The distribution of the specificities in different inbred strains is shown in Table 24-10. Kappa occurs only in strain YBR/HeHa, Iota in all strains tested except C3H/St, C58, and F/St. Both specificities segregate independently of H-2.
Popp and Popp ( 1964), using an antiserum produced in C3H mice against RFM tissues and subsequently absorbed with tissues of strain A.CA, have identified a specificity which occurs only in strain RFM of 13 strains tested. The specificity is not related to H-2. It is not confined to erythrocytes, and hence may be of significance for histocompatibility.
Singer et al. ( 1964) have reported a blood group locus Ea-a, with alleles Ea-1a, Ea-ab, and Ea-ao, detected in wild house mice. Antibodies are induced by the injection of erythrocytes, and unlike other alloantibodies in mice behave as saline or complete agglutinins. Four phenotypes are demonstrable, A, B, AB, and O. All of 13 inbred strains tested are O. The locus is distinct from H-2.
Besides the histocompatibility loci so far described, five others have been identified by the application of the F1 test to lines congenic with C57BL/10 ( Snell and Bunker, 1965; Table 24-8). These loci have tentatively been assigned the symbols H-7, H-8, H-9, H-10, and H-11. The distribution of allele H-7a in various inbred strains has been determined by the F1 test ( Table 24-10). The distribution suggests a difference between H-7 and the blood group loci H-5 and H-6 (compare, e.g., strains DBA/1 and 129), but this evidence is subject to the same qualifications as are other comparisons with these two loci. Amos et al. ( 1963) tested, with their H-5 typing antiserum, the congenic lines that identify H-8 and H-11 and found that they react like C57BL/10. This establishes a presumption that H-8 and H-11 are distinct from H-5, the presumption being particularly strong in the case of H-11 because of the key role of strain 129 in the identification of both H-5 and H-11. Aside from the results of these few tests, the nonidentity of these presumed new loci with H-5 and H-6 is unproven.
The loci, the CR strains that identify them, and some results with tumor and skin allografts are given in Table 24-11. The table gives similar data for H-1, H-2, H-3, and H-4. The principal point of interest is the marked differences in the strength of the barrier to allografts imposed by the congenic strain pairs. This was noted in an earlier discussion of the table. Presumably this points to comparable differences in the "strength" of the different loci, but there is a distinct possibility that different allelic combinations within the same locus would alter the results. (See for example the different H-1 combinations in Table 24-11). On the basis of evidence at present available, the comparative immunogenic strength of the nine loci listed in the table is, in descending order: H-2, H-1, H-3, H-7, H-8, H-4 (but this is relatively "strong" in the reciprocal direction), H-9, H-10, H-11 (but the H-11 allele present in strain B10.D2(55N) would place this locus in a higher position).
TWO THYMIC ALLOANTIGENS
Two alloantigens, probably genetically distinct, have been identified in the thymus of mice.
The TL antigen is determined by a locus Tla in linkage group IX about two crossover units from H-2 ( Figure 24-4). It is present in thymus, as demonstrated by the capacity of thymus to absorb appropriate antisera, and in certain leukemias as demonstrated by a cytotoxic test. (Hence the designation TL = thymus leukemia.) It is not present in liver, spleen, lymph nodes, or red cells. The strain distribution is shown in Table 24-10. It is present in the thymus of strains A and C58, lacking in the thymus of strains AKR, BALB/c, C3H/He and C57BL/6. Curiously, it is present in some leukemias arising in strains that lack it. It has been possible to produce reactive antisera only when leukemias possessing the antigen are used as the immunizing tissue, not when thymus is so employed. The TL antigen may not affect histocompatibility; C57BL/6 mice with a high titer of TL alloantibody did not reject leukemias possessing the antigen ( Old et al., 1963; Boyse et al., 1963, 1964; Boyse et al., 1965).
Reif and Allen ( 1964) have discovered a second and possibly distinct thymic antigen which occurs in two antithetical forms. θ-AKR is present in strains AKR and RF, lacking in all other strains tested. θ-C3H is lacking in AKR and RF, present in all other strains tested ( Table 24-10). The antigens are demonstrated by the cytotoxic action on thymic lymphocytes of alloantisera prepared in H-2-compatible strains AKR and C3H. The antigens are present in brain as well as thymus, lacking or present in very low concentrations in other tissues tested. Genetic determination has not been demonstrated, but the distinct strain distribution certainly points to determination by a single locus. There is no evidence as to whether or not the antigens affect histocompatibility.
THE X-LINKED HISTOCOMPATIBILITY FACTOR
Bailey ( 1963) has demonstrated the presence of a histocompatibility factor on the X chromosome of mice. The evidence is summarized in Table 24-16. Male F1 hybrids between strains C57BL and BALB/c bearing an XB (X from strain C57BL) rejected grafts with and XC (X from strain BALB/c) regardless of the sex of the donor and those bearing an XC rejected grafts with an XB. Grafts of all other genotypes were accepted. (Male to female grafts were not made.) Rejection was slow, many grafts surviving beyond 35 days, and in the combination CBF1 to BCF1, 14 of 36 grafts had not been rejected at 63 days when the test was terminated. The X locus thus determines a weak immunogen. Perhaps it is especially weak, since the grafts used were tail skin placed on the tail, and there is some reason to believe that grafts of this type are particularly responsive to allogeneic differences (Bailey, personal communication). There is no means at present of telling whether the effect is due to one locus or several, and no evidence as to the number and distribution of alleles at this locus or these loci beyond the fact that strains C57BL and BALB/c must have different alleles.
THE Y-LINKED HISTOCOMPATIBILITY
In 1955 Eichwald and Silmser reported that grafts of skin made from male to female within an inbred strain are rejected, whereas grafts from male to male, female to female, and female to male are accepted. This sex-associated incompatibility has been widely studied. Since the Y chromosome of the X/Y male donor is foreign to the X/X female recipient in male-to-female grafts, and in this one combination only, the phenomenon has been generally interpreted as due to a Y-linked histocompatibility gene (or genes).
The rejection due to the Y factor is weak and shows pronounced strain differences. The rejection is entirely consistent in strain C57BL, but mean rejection time has varied from 26 to 45 days in the hands of different investigators. Table 24-11 includes the results of one typical experiment. In other strains, e.g., A and BALB/c, only a fraction of the grafts are rejected, while in strain CBA two extensive experiments failed to produce any rejections at all ( Eichwald et al., 1958; Klein and Linder, 1961; Zaalberg, 1959). (But at least one author has reported some rejections in this combination. See Hauschka et al., 1961, for a more complete summary of published results.
In strain C57BL, in which rejections occur regularly, a second graft is rejected more rapidly than a primary one. Thus 13 C57BL females that had rejected an initial male graft in an average of 26 days, rejected a second graft in an average of 13 days. Also testosterone treatment or castration of the female hosts failed to alter rejection time ( Eichwald et al., 1958). The sex-determined rejection thus behaves, as do other rejections of allografts, like an immune phenomenon incited by alloantigenic differences between donor and host.
If the rejection of male-to-female skin grafts is, in fact, due to an antigenic difference between the sexes, the obvious place to look for the genetic determinant of the difference is on the Y chromosome. A Y-linked histocompatibility gene would explain the results, at least so far as strain C57BL is concerned. A Y-linked gene, however, as Fox ( 1959) has pointed out, is not the only possible explanation. Important modifications of the phenotype, including, apparently, differences in the presence or absence of antigens, can be brought about by modifications in the proportions or balance of the various chromosomes. Studies with Drosophila, for example, provide precedents for postulating that the male antigen is determined, not by a Y-linked factor, but by the presence of one rather than two X's.
Celada and Welshons ( 1963) have carried out experiments which discriminate between these alternatives. Their results conform the presence of a histocompatibility factor on the Y chromosome. The experiments made use of an exceptional stock of mice in which some animals have the genetic constitution of X/O (one X and no Y) ( Welshons and Russell, 1959) and of a single exceptional mouse of the genotype XXY. The X/O animals are fertile females; the XXY mouse was a male. Since these animals were not from inbred stocks, the role of the Y could not be tested by direct male-to-female skin grafts. Instead, the presence or absence of the male antigen was demonstrated indirectly by the capacity of tissues bearing it to immunize. Females were immunized with spleen cells and then challenged with an isogenic male graft. Another unusual feature of the system was the use, not of skin, but of male spleen cells from mice presensitized to rat erythrocytes as the challenge. Reduced production of anti-erythrocyte antibody was taken as evidence of preimmunization. The method involves other interesting details, but these need not concern us here. The point of genetic significance is that XXY tissue did immunize and X/O tissue did not. Evidently the male antigen can safely be attributed to the presence of the Y rather than the absence of a second X. It is truly a Y-linked histocompatibility factor (or factors). These experiments also show that the Y-determined antigen is carried by spleen cells as well as by skin.
While the existence of a Y-linked factor may be regarded as established, the absence of male-to-female graft rejection in strain CBA suggests that the factor may be absent in this strain. But other explanations of this curious strain difference have been proposed. At least four alternatives are possible: (1) The Y of strain CBA lacks the male factor; (2) the male factor is on the X as well as the Y of this strain; (3) CBA's have the Y factor, but they also have an autosomal factor which produces an identical antigen ( Michie and McLaren, 1958); (4) the CBA's are less effective than the C57BL's in mounting an immune response against weak antigens or at least against this particular antigen. Tests have now been reported which discriminate quite effectively between these alternatives.
Zaalberg ( 1959) tested C57BL, CBA, and F1 hybrid male skin in F1 male and female hybrids. His most critical results are given in Table 24-17. The rejection of CBA male skin proves that the CBA Y chromosome carries the male factor, and that the CBA X lacks it. This result also proves that CBA does not carry an autosomal replica of the Y factor or at least not a replica that acts as a dominant. Since all known histocompatibility factors in the mouse are dominant (or more precisely codominant), including the Y factor, the postulate of a recessive autosomal replica of the Y factor is improbable. This one simple experiment thus rules out the first three of the four proposed explanations. Klein and Linder ( 1961) obtained the same sort of result in the critical combination of grafts from CBA males to (CBA x C57BL)F1 females. It must be added that three out of 23 of Zaalberg's CBA male to F1 female grafts were accepted, a problem we shall return to later. Linder and Klein obtained 100 per cent rejection.
Zaalberg's tests establish one other point. Forty of 40 C57BL male to (C57BL x CBA)F1 male grafts were accepted. Since donor and recipient in this combination have Y chromosomes from strains C57BL and CBA, respectively, the histocompatibility antigens determined by the two Y chromosomes must be identical. The genetic formulas used to designate donor and recipient in Table 24-17 should help one to visualize this situation.
Experiments by Billingham and Silvers ( 1960), utilizing the capacity of neonatally injected marrow to induce tolerance to subsequent grafts of the same genotype, confirm the presence of the male factor in strain CBA. C57BL females made tolerant to CBA male tissue accepted C57BL male skin. CBA males must carry the antigen that normally causes C57BL male skin to be rejected. Similar results were obtained with strains A, AU, and C3H. Controls injected neonatally with female tissue were not rendered tolerant.
We are left then, as the most likely explanation of the absence of male-to-female graft rejection in strain CBA, with the postulate that CBA does not mount an effective immunological response against the male antigen. Differences in immunological response in different strains of mice are well established, and Klein and Linder ( 1961) have summarized data which suggest a correlation between the immunological capacity of several strains and their tendency to show male-to-female graft rejection.
These authors have also tested CBA male skin in females from a backcross produced by crossing (CBA female x C57BL male)F1 males to CBA females. In a test thus designed, all sex chromosomes of both donor and recipient are of CBA origin. Of 110 backcross mice grafted, 85 rejected their grafts. The rejection times of the sloughed grafts were more varied than in grafts from CBA males to F1 females. These results fit the postulate that the CBA Y chromosome carries the same male factor as the Y of strain C57BL and that the difference between the strains resides in autosomal modifying factors, presumably factors that affect the organs responsible for the immune response. The authors tentatively suggest that two dominant autosomal genes are involved, but interpretation of multiple-factor situations is difficult, and the number of factors should be regarded as uncertain at the present time. The results do not fit the possibility discussed above that the exceptional response of strain CBA is due to a recessive autosomal replica of the Y factor. This would require 50 per cent rejected grafts in the backcross instead of the 77 per cent observed.
It remains to consider the explanation of those cases, exemplified by strain A, in which some male-to-female isografts are accepted and others rejected. This situation is examined in the next section.
PENETRANCE OF HISTOCOMPATIBILITY
In a number of the studies cited, donor and recipient have differed one from the other at a weakly immunogenic locus. The results have been ambiguous in the sense that some but not all of the grafts have been rejected. Thus only 23 of 39 grafts of strain A skin made from males to females were rejected at the end of 5 months ( Eichwald et al., 1958). Similar results are common in the case of tumor allografts; some animals of a given genotype are killed and others survive ( Table 24-11). Since these grafts are made between inbred animals, the results may be puzzling to persons to whom inbreeding has become synonymous with uniformity. Actually, results of this sort are no novelty. Geneticists have known for years examples of variation in inbred strains, sometimes of a very striking nature (see Wright, 1934, for the classic example). They also know a great variety of mutant genes which, both on inbred and noninbred backgrounds, may fail to show 100 per cent expression. To describe this last phenomenon they have coined the term "penetrance," defined as the percentage of animals in a given population in which a given gene is expressed, or in which phenotype corresponds to genotype (e.g., the penetrance of the Y chromosome factor in A male to A female skin grafts is 59 per cent).
While results of this sort may at first seem puzzling, a little consideration will show that they have an obvious explanation. All traits, even those most directly related to gene action, are determined at least in part by environment. Imperfect penetrance is thus merely a reflection of the interaction of environment with genotype. In noninbred animals, an important part of the "environment" of any given gene is the rest of the gene complex. In inbred animals, the gene complex is uniform, but its expression may be modified by any or all of a great variety of forces to which it is exposed, forces which begin at conception and do not terminate till death. Variation of a given trait within an inbred strain thus becomes a direct measure of the role of environment in determining that trait. Our knowledge of the environmental forces influencing tissue allografts is far from complete, but the role of certain factors is well established. One of the more important is age of the host, with age of the donor in some cases also playing a role. A typical experiment with skin grafts, reported by Mariani et al. ( 1959) is summarized in Table 24-18. The percentage of success of male-to-female skin grafts made in strain A was 75 when donors and hosts were both young (6 and 5 weeks of age, respectively), but was 0 per cent when both were fully mature (31 and 39 weeks). Other age combinations gave intermediate results. The role of the age of the host is well established. Maximum resistance is reached at about 12 weeks; presumably this coincides with the attainment of full immunological maturity (see Snell, 1958a, for summary). The effect of age of donor is less striking, but the relatively prolonged survival of immature skin is well documented (see e.g., Cannon et al., 1954; Billingham and Silvers, 1964).
There are a number of other known environmental factors that influence the outcome of grafts, e.g., the tissue employed, the site in which it is placed, and prior immunization of the recipient. Even if all these known factors are controlled, some variation persists in the outcome of allografts, especially allografts where the genetic disparity between donor and host is marginal. A translocation of the Y factor to an autosome has been invoked to explain results of this type obtained with male-to-female grafts ( Hauschka and Holdridge, 1962). Possibly such explanations are justified by the facts in a few instances. However, imperfect penetrance of genes on an inbred background due to shifts in minor and largely obscure environmental factors is well known for a variety of genes and there is no reason to doubt that it applies to histocompatibility genes also. A weak level of resistance, a delicate balance between the mechanisms of immunity and tolerance, and fluctuations in minor environmental factors are quite sufficient to account for these cases of imperfect penetrance. What the minor environmental factors are we can only guess, but the state of the hair-growth cycle in the graft, the amount of blood in the graft, the site of the graft relative to host blood vessels, minor technical fluctuations, the state of the estrus cycle of the host, and the health and infective state of the host are some of the possibilities.
Of course, if one were able to measure directly the presence of the histocompatibility alloantigens and use this as the measure of gene penetrance, the penetrance of histocompatibility genes would doubtless be found to be almost, if not quite, 100 per cent.
EXCEPTIONS TO THE LAWS OF TRANSPLANTATION
That there are exceptions to the some of the five laws of transplantation described in an earlier section should now be apparent. The commonest exceptions concern law 2 which asserts the incompatibility of all allografts. There is some reason to suppose that allografts are, in fact, almost always resisted, except perhaps in the case of certain tissues (e.g., cartilage), or tissues placed in certain "privileged sites" (e.g., the anterior chamber of the eye), but they are certainly not always rejected. Exceptions are very common with tumors, especially across non- H-2 barriers. Generally speaking, the tendency of tumors to grow progressively in foreign hosts increases with the number of transplant generations through which they have been carried. "Antigenic simplification" and increased capacity to survive the immunological attack of the host have been suggested as explanations. Probably the latter is the more important. Some tissues, e.g., ovary, grow quite easily across weak histocompatibility barriers ( Linder, 1961; Snell and Stevens, 1961). Even skin, probably the most sensitive of all histocompatibility indicators, at least in the absence of prior immunization sometimes transgresses the weaker barriers.
Law 3, which asserts the compatibility of tissues grafted from an inbred parent to its F1 hybrid, was long regarded as universally valid, but a major exception has been found (Cudkowicz and Stimpfling, 1964a, 1964b; 1965a, 1965b). The phenomenon is noted when lymphoid tissues of the genotype H-2b/ H-2b are grafted to recipients heterozygous for H-2b and some other H-2 allele. By the use of alleles derived by crossing over within the H-2 locus, it has been shown that the effect is controlled by a factor which is either a part of or close to the D region of the H-2 locus ( Figure 24-4). It has been suggested that an antigenic specificity normally produced by allele H-2b is not produced when H-2b is combined with another H-2 allele. Heterozygotes can therefore react against H-2b homozygotes. Since, however, the phenomenon is noted only with grafts of lymphoid tissues and not, for example, with skin, the postulated specificity must be formed only in such tissues. Also there is some reason to doubt that rejection of H-2b/ H-2b marrow by F1 hybrids is an immune phenomenon. The test for successful marrow transplantation employed by Cudkowics and Stimpfling is carried out in irradiated hosts. C3H hosts irradiated with 1,000 R show no resistance to C57BL/10 marrow by this test despite the "strong" H-2 difference, yet in (C57BL/10 x C3H)F1 hosts receiving the same radiation dose the hybrid resistance is fully manifested (Cudcowicz and Stimpfling, 1964a). On the other hand hybrids pretreated with marrow show an increased resistance suggestive of immunity in genetic combinations expected to show the hybrid effect, and hybrids pretreated with spleen show a reduced resistance suggestive of tolerance ( Cudkowicz and Stimpfling, 1964c).
The rejection of 5 x 105 cells of H-2b/ H-2b marrow by heterozygous recipients noted by Cudcowicz and Stimpfling occurs within 5 days. When 107 nucleated cells are used, repopulation of the recipient usually though not always occurs. Popp and Cudkowicz ( 1965) have shown that the failures under these conditions are not related to H-2b, though they are dependent on the use of heterozygous recipients.
Other but much less striking examples of hybrid resistance have been reported with transplantable sarcomas and carcinomas in genotypes not confined to H-2b ( Hellström, 1964). Studies of this sort have led to the detection of the interesting phenomenon of syngeneic preference in which cells in tissue culture are damaged by contact with histoincompatible cells or even histoincompatible alloantigens ( Hellström et al., 1964; Möller, 1965). If hybrid resistance, at least in some instances, is indeed a manifestation of syngeneic preference, then the role of the F1 hybrid hosts is to provide an environment for transplants in which intimate contact can occur between cells which are genetically disparate and probably disparate with respect to the alloantigens of their cell membranes, and yet in which no ordinary immunological reaction can mask the resulting cell damage.
ARE THERE "HYBRID ANTIGENS" IN THE MOUSE?
In rabbits and some other species, animals heterozygous at certain blood group loci have been shown to possess an alloantigen or alloantigens not determined by the alleles of these loci acting alone. A search for similar "hybrid antigens" in mice gave negative results. Martinez et al. ( 1959) rendered strain A mice tolerant to C3H alloantigens, and then grafted them simultaneously with C3H and (A x C3H)F1 skin. Both grafts were accepted. The same results were obtained in the reciprocal combination. Had heterozygosity at any of the histocompatibility loci by which strains A and C3H differ led to the formation of alloantigens not present in the homozygotes, the hybrid grafts should have been rejected. The test of course gives no information about loci at which the strains used are alike or about alloantigens confined to tissues other than skin.
NUMBER OF HISTOCOMPATIBLITY LOCI
In an earlier section we discussed the formulas by which the percentage of susceptible animals in the segregating generations of a cross is related to the number of segregating histocompatibility loci, but we deferred actual attempts to estimate the number of loci. We are now in a better position to evaluate such data as are available. There are at least three and perhaps four factors that would lead to underestimation of the number of loci: (1) Incomplete penetrance would increase the proportion of survivors and therefore lower the estimate. This is particularly true of tumor transplants but we have seen that it can apply to skin grafts also. (2) Linked loci would tend to behave like a single locus and would add less to the estimate of gene number than would the same loci if unlinked. Linked histocompatibility loci are already known and more will certainly be discovered. (3) If the strains crossed carry the same allele at any locus, this locus would make no contribution to the estimate of gene number. How often this is likely to occur depends on the number of alleles at different loci. At present this is largely an immeasurable factor. (4) It may well be that, because of an unequal distribution of alloantigens in the different tissues, some types of grafts fail to detect loci that would be demonstrable were other tissues employed.
There are no comparable factors that would tend to lead to overestimation of the number of loci. However, in the two tests with skin grafts summarized in Table 24-2 some use was made of male donors with female recipients. This could lead to rejections due to the Y chromosome factor and to an elevation of the estimate of locus number. An extenuating circumstance is that the strains used happen to be ones in which the Y chromosome effect is particularly weak. Strains CBA and DBA/2 practically do not show it at all. Perhaps the results can be taken at their face value.
These tests led to estimates of approximately 13 and 15 loci, respectively. In the light of the sources of error mentioned above, these are clearly minimum figures. It should also be remembered that in Barnes and Krohn's tests, all grafts were sloughed by 200 days. While this implies that some of the loci acting are very weakly immunogenic, it leads to a necessary but indeterminate elevation of estimated locus number.
Chai and Chiang ( 1963) devised a mathematical method for estimating histocompatibility gene number from the percentage acceptance of grafts exchanged between sibs in a partially inbred strain. Using data from the 14th to 19th generations of brother-sister inbreeding of strain LG, they arrived at an estimate of 18 histocompatibility loci. If there are selective forces favoring heterozygosity at histocompatibility loci, this would tend to make this figure an overestimate. But the other factors already enumerated would tend to make it an underestimate. The figure would not include the X and Y chromosome loci.
Bailey and Mobraaten ( 1964), using the same genetic device but grafts of tail skin to the tail rather than the more familiar thoracic grafts, arrived at a figure of 32 loci. In a second test using similar grafts but animals of an N5 generation, the same authors found evidence for 29 loci. The high estimates from these last studies possibly reflect an unusual responsiveness of the tail skin method to allogeneic differences (Bailey, personal communication).
Quite evidently estimates of about 14 or 15 histocompatibility loci which have frequently appeared in the literature are substantial underestimates. Quite evidently also the number of loci which we call histocompatibility loci is a function of the methods we use to detect them.
MUTATIONS AT HISTOCOMPATIBILITY LOCI
There is considerable evidence that, with passage of time, occasional minor or "weak" histocompatibilities arise within inbred strains, presumably due to mutation. Linder ( 1963) performed 600 skin grafts between sublines of CBA separated 0 to 18 generations. (The number of generations was taken as the sum of the generations separating donor and recipient from their common ancestral brother-sister pair.) There were no rejections in grafts between 280 pairs separated zero to eight generations, three delayed rejections (82 to 100 days) in 320 similar grafts in generations 10 to 18. Four additional grafts in the latter group showed signs of incipient rejection but were nevertheless retained. Linder was cautious in interpreting the histoincompatibility thus revealed as due to mutation, but since strain CBA had passed through some 120 generations of brother-sister inbreeding prior to the beginning of the tests, mutation seems a more likely explanation than continued segregation. Billingham et al. ( 1954) had previously reported signs of incipient rejection in grafts between sublines of A and CBA, and Snell and Stevens ( 1961) found that sublines 6 and 10 of strain C57BL, separated by some 25 years after prolonged inbreeding, could be easily differentiated with a transplantable tumor if the hosts were given prior immunization with donor tissue. Godfrey and Searle ( 1963) noted no rejections in 82 grafts of tail skin made between sublines of C3H which had been subjected to prolonged gamma irradiation. Donor and host were separated by an average of about 34 generations.
Major differences at the H-2 locus have also been reported as appearing within inbred strains, but the circumstances of discovery of these variants were such as to make it difficult to rule out contamination. In one case a variety of other differences has been shown, by several methods, to accompany the H-2 difference (see, e.g., Snell, 1958b).
DISTRIBUTION OF HISTOCOMPATIBILITY ALLOANTIGENS IN CELLS AND TISSUES
The distribution of the histocompatibility alloantigens in cells and tissues is a complex problem about which little is known. Yet it is an important problem in transplantation studies, since the potential for histocompatibility of a particular tissue obviously resides in the alloantigens which it carries.
Much of the information so far gathered on tissue distribution concerns three loci, H-2, H-5, and H-6, and is based on the capacity of tissue homogenates to absorb the appropriate antibodies. Basch and Stetson ( 1962) studied the tissue distribution of the H-2 alloantigen and Amos et al. ( 1963) of H-2, H-5, and H-6. Some of the results of these authors are summarized in Table 24-19. The figures in the table indicate the relative, not the absolute, concentration of the different antigens. It will be seen that the two different studies gave essentially identical results concerning the tissue distribution of the H-2 substance, but that when different alloantigens were compared, the distributions showed striking differences. Thus high concentrations of H-2 are found in the spleen and liver; of H-5 in the kidney, red cells, and testis; of H-6 in the gut, red cells, testis, and brain. While both spleen and liver are relatively rich in H-2, the properties of H-2 appear to differ in these two organs. The H-2 substance in liver is more easily sedimented than that in spleen, and it is less effective in inducing antibody formation, particularly following a single injection of lyophilized tissue ( Pizarro et al., 1963).
The Y-determined antigen, like H-2, H-5, and H-6, probably occurs in a variety of tissues, since male-to-female interstrain grafts of a number of tissues besides skin are rejected (see, for example, Gittes and Russell, 1961). Katsh et al. ( 1964) have shown that there is accelerated rejection of male skin in isogeneic females injected with 1 million sperm and delayed rejection (tolerance) in females injected with 8 million sperm. Apparently the Y antigen is present on sperm, in fact a comparison of the effectiveness of sperm as compared with a preparation of spleen cells in producing immunity and tolerance suggests that it is present on sperm in particular abundance.
As Billingham et al. ( 1956) point out, the capacity of neonatally injected spleen to induce tolerance to a subsequent skin graft from the same donor strain proves that all the transplantation alloantigens of the latter tissue must be present on the former. It does not necessarily follow, however, that the relative concentration of the antigens is the same in both tissues.
As to the intracellular distribution of the histocompatibility alloantigens, the best available evidence points to an association with the cell membrane. The evidence is quite firm in the case of H-2 and of those other alloantigens ( H-1, H-5, H-6) which are demonstrable by red-cell agglutination, but is of an uncertain nature in all other cases. The H-2 alloantigen, at least, is probably associated with the membranes of the endoplasmic reticulum as well as with the surface membrane. For references and further details, see Snell ( 1963, 1964a).
SUMMARY AND CONCLUSIONS
In summary, the compatibility of tissue grafts is governed by 15 or more loci called histocompatibility loci. Fifteen is a minimum estimate; the actual number may be substantially more than this. These loci are widely distributed throughout the chromosomes. Histocompatibility genes have so far been identified on the X and Y, and in linkage groups I, V, and IX.
Histocompatibility genes probably act as such because their end products are alloantigens, i.e., possess the property of inciting an immune response when transferred via a graft from an individual possessing them to an individual lacking them. Even though all histocompatibility genes may determine alloantigens, the reverse proposition, that all alloantigens play a role in histocompatibility, is probably not true. For example, a gene whose alloantigenic end product is confined to the fluid components of the blood will presumably not affect histocompatibility.
Histocompatibility loci differ in their immunogenicity or "strength." Some impose a much more powerful barrier to transplants than do others. The H-2 locus is seldom transgressed even by transplants of most tumors; the Y-chromosome locus is essentially without effect on some genetic backgrounds even when tested by such a sensitive agency as skin grafts. These properties of the loci presumably relate to the character, quantity, or location of the alloantigen which each determines.
For a few of the histocompatibility loci of the mouse, there is rather conclusive evidence that their end product is a component of the cell membrane. It is tempting to speculate that this is true of most or all of the 15 or more such loci, excepting, perhaps, some of those with particularly weak effects. The histocompatibility loci would then be the loci whose normal function is to produce the chemical building blocks from which the cell membrane is formed. While such evidence as is available indicates a wide distribution in the different tissues of the various histocompatibility alloantigens, the relative concentrations of these substances may vary greatly. As to the developmental factors which determine these variations in concentration we have no knowledge, but in some way the different H loci contribute more or less to the cell — more specifically, perhaps, to the cell membrane — according to the tissue in which they operate. They thereby contribute to the individuality of tissues as well as to the individuality of the organism as a whole.
1The writing of this chapter was supported in part by Public Health Service Research Grant CA 01329 from the National Cancer Institute and by a Public Health Service Research Career Award K6 CA 21784. 2
2Subsequent to writing this chapter, 5 and 31 were found in combination in allele H-2a (Shreffler, unpublished data) and possibly in H-2r (Snell, unpublished data).
LITERATURE CITED
Allen, S.L. 1955a.
H-2f, a tenth allele at the histocompatibility-2 locus in the mouse as determined by tumor transplantation.
Cancer Res. 15: 315-319.
See also
MGI.
Allen, S.L. 1955b.
Linkage relations of the genes histocompatibility-2 and fused tail, brachyury and kinky tail in the mouse as determined by tumor transplantation.
Genetics 40: 627-650.
See also
MGI.
Amos, D.B. 1958.
Genetic studies on tumor immunity: an analysis of C3H sublines and their tumors.
Ann. N.Y. Acad. Sci. 71: 1009-1021.
See also
PubMed.
Amos, D.B. 1959. Some iso-antigenic systems of the mouse. Proc. 3rd Can. Cancer Conf. 3: 241-258.
Amos, D.B. 1962.
Isoantigens of mouse red cells.
Ann. N.Y. Acad. Sci. 97: 69-82.
See also
PubMed.
Amos, D.B., P.A. Gorer, and Z.B. Mikulska. 1955a.
The antigenic structure and genetic behavior of a transplanted leukosis.
Brit. J. Cancer 9: 209-215.
See also
PubMed.
Amos, D.B., P.A. Gorer, and Z.B. Mikulska. 1955b.
An analysis of an antigenic system in the mouse (the H-2 system).
Proc. Roy. Soc. B 144: 369-380.
See also
PubMed.
Amos, D.B., M. Zumpft, and P. Armstrong. 1963.
H-5.A and H-6.A, two mouse isoantigens on red cells and tissues detected serologically.
Transplantation 1: 270-283.
See also
MGI.
Bailey, D.W. 1963.
Histoincompatibility associated with the X chromosome in mice.
Transplantation 1: 70-74.
See also
MGI.
Bailey, D.W., and L.E. Mobraaten. 1964. Estimates of the number of histocompatibility loci at which the BALB/c and C57BL/6 strains of mice differ. Genetics 50: 233. (Abstr.)
Barnes, A.D., and P.L. Krohn. 1957.
The estimation of the number of histocompatibility genes controlling the successful transplantation of normal skin in mice.
Proc. Roy. Soc. B 146: 505-526.
See also
PubMed.
Basch, R.S., and C.A. Stetson. 1962.
The relationship between hemagglutinogens and histocompatibility antigens in the mouse.
Ann. N.Y. Acad. Sci. 97: 83-94.
See also
PubMed.
Berrian, J.H., and C.F. McKhann. 1960a. Strength of histocompatibility genes. Ann. N.Y. Acad. Sci. 87: 106-111.
Berrian, J.H., and C.F. McKhann. 1960b.
Transplantaation immunity involving the H-3 locus: graft survival times.
J. Nat. Cancer Inst. 25: 111-123.
See also
PubMed.
Billingham, R.E., L. Brent, and P.B. Medawar. 1956. Quantitative studies on tissue transplantation immunity. III. Actively acquired tolerance. Phil. Trans. Roy. Soc. B 239: 357-414.
Billingham, R.E., L. Brent, P.B. Medawar, and E.M. Sparrow. 1954.
Quantitative studies on tissue transplantation immunity. I. The survival times of skin homografts exchanged between members of different inbred strains of mice.
Proc. Roy. Soc. B 143: 43-58.
See also
PubMed.
Billingham, R.E., and W.K. Silvers. 1960.
Studies on tolerance of the Y chromosome antigen in mice.
J. Immunol. 85: 14-26.
See also
MGI.
Billingham, R.E., and W.K. Silvers. [ed.] 1961. The Transplantation of Tissues and Cells. Wistar Institute Press, Philadelphia. 149 p.
Billingham, R.E., and W.K. Silvers. 1963.
Sensitivity to homografts of normal tissues and cells.
Annu. Rev. Microbiol. 17: 531-564.
See also
PubMed.
Billingham, R.E., and W.K. Silvers. 1964.
Studies on homografts of foetal and infant skin and further observations on the anomalous properties of poch skin grafts in hamsters.
Proc. Roy. Soc. B 161: 168-190.
See also
PubMed.
Boyse, E.A., L.J. Old, and S. Luell. 1963. Antigenic properties of experimental leukemias. II. Immunological studies in vivo with C57BL/6 radiation-induced leukemias. J. Nat. Cancer Inst. 31: 987-995.
Boyse, E.A., L.J. Old, and S. Luell. 1964.
Genetic determination of the TL (thymus-leukaemia) antigen in the mouse.
Nature 201: 779.
See also
MGI.
Boyse, E.A., L.J. Old, and E. Stockert. 1965. TL: a leukemia-specific antigen determined by the genome of the leukemic cell. Symp. Immunopathol. (Monte-Carlo). (In press.) (Abstr.)
Brent, L., P.B. Medawar, and M. Ruszkiewicz. 1961.
Serological methods in the study of transplantation antigens.
Brit. J. Exp. Pathol. 42: 464-477.
See also
PubMed.
Brondz, B.D., and I.K. Yegorov. 1964.
Antigenic structure of locus H-2 of the mouse strains CC57BR and CC57W.
Folia Biol. (Prague) 10: 90-93.
See also
PubMed.
Cannon, J.A., R.A. Weber, and W.P. Longmire, Jr. 1954.
Factors influencing the survival of successful skin homografts in the chicken. I. Effects of varying age of donor and recipient.
Ann. Surg. 139: 468-472.
See also
PubMed.
Castermans, A., and A. Oth. 1959.
Transplantation immunity: separation of antigenic components from isolated nuceli.
Nature 184: 1224-1225.
See also
PubMed.
Celada, F., and W. J. Welshons. 1963.
An immunogenetic analysis of the male antigen in mice utilizing animals with an exceptional chromosome constitution.
Genetics 48: 139-151.
See also
PubMed.
Chai, C.K., and M.S.M. Chiang. 1963.
A method of estimating the number of histocompatibility loci in a sib-mating mouse population.
Genetics 48: 1153-1161.
See also
PubMed.
Cloudman, A.M. 1932. A comparative study of transplantability of eight mammary gland tumors arising in inbred mice. Amer. J. Cancer 16: 568-630.
Counce, S., P. Smith, R. Barth, and G.D. Snell. 1956.
Strong and weak histocompatibility differences in mice and their role in the rejection of homografts of tumors and skin.
Ann. Surg. 144: 198-204.
See also
PubMed.
Cudkowicz, G., and J.H. Stimpfling. 1964a.
Deficient growth of C57BL marrow cells transplanted in F1 hybrid mice: association with the histocompatibility-2 locus.
Immunology 7: 291-305.
See also
PubMed.
Cudkowicz, G., and J.H. Stimpfling. 1964b.
Hybrid resistance to parental marrow grafts: association with the K region of H-2.
Science 144: 1339-1340.
See also
MGI.
Cudkowicz, G., and J.H. Stimpfling. 1964c.
Induction of immunity and of unresponsiveness to parental marrow grafts in adult F1 hybrid mice.
Nature 204: 450-453.
See also
PubMed.
Cudkowicz, G., and J.H. Stimpfling. 1965a.
Hybrid resistance controlled by H-2 region: correction of data. Science 147: 1056.
See also
PubMed.
Cudkowicz, G., and J.H. Stimpfling. 1965b.
Lack of expression of parental isoantigen(s) in F1 hybrid mice, p. 144-148.
In: Proc. X Cong. Int. Soc. Blood Transfusion. S. Karger, Basel.
See also
PubMed.
Dhaliwal, S.S. 1964. Histocompatibility variations in mouse tumor cells and embryonic tissue with the use of C57BL/10Sn strain and its isogenic resistant strains. J. Nat. Cancer Inst. 32: 1001-1022.
Eichwald, E.J., and C.R. Silmser. 1955.
(Note without title.)
Transplant. Bull. 2: 148-149.
See also
PubMed.
Eichwald, E.J., C.R. Silmser, and I. Weissman. 1958.
Sex-linked rejection of normal and neoplastic tissue. I. Distribution and specificity.
J. Nat. Cancer Inst. 20: 563-575.
See also
PubMed.
Fox, A.S. 1959.
Genetic determination of sex-specific antigens.
J. Nat. Cancer Inst. 23: 1297-1311.
See also
PubMed.
Gittes, R.F., and P.S. Russell. 1961.
Male histocompatibility antigens in mouse endocrine tissues: functional and histologic evidence.
J. Nat. Cancer Inst. 26: 283-303.
See also
PubMed.
Godfrey, J., and A.G. Searle. 1963. A search for histocompatibility differences between irradiated sublines of inbred mice. Genet. Res. 4: 21-29.
Gorer, P.A. 1937. The genetic and antigenic basis of tumor transplantation. J. Pathol. Bacteriol. 44: 691-697.
Gorer, P.A. 1938. The antigenic basis of tumor transplantation. J. Pathol. Bacteriol. 47: 231-252.
Gorer, P.A. 1942. The role of antibodies in immunity to transplanted leukemia in mice. J. Pathol. Bacteriol. 54: 51-65.
Gorer, P.A. 1956.
Some recent work in tumor immunity.
Adv. Cancer Res. 4: 149-186.
See also
PubMed.
Gorer, P.A. 1959. Some new information on H-2 antigens in mice, p. 25-30. In F. Albert and P.B. Medawar [ed.] Biological Problems of Grafting. Blackwell Scientific Publications, Ltd., Oxford.
Gorer, P.A., S. Lyman, and G.D. Snell. 1948. Studies on the genetic and antigenic basis of tumor transplantation. Linkage between a histocompatibility gene and "fused" in mice. Proc. Roy. Soc. B. 135: 499-505.
Gorer, P.A., and Z.B. Mikulska. 1954.
The antibody response to tumor inoculation. Improved methods of antibody detection.
Cancer Res. 14: 651-655.
See also
PubMed.
Gorer, P.A., and Z.B. Mikulska. 1959.
Some further data on the H-2 system of antigens.
Proc. Roy. Soc. B 151: 57-69.
See also
MGI.
Graff, R.J., W.H. Hildemann, and G.D. Snell. 1965.
Histocompatibility genes of mice. VI. Allografts in mice congenic at various non-H-2 histocompatibility loci.
Transplantation 4: 425-437.
See also
PubMed.
Hauschka, T.S., S.T. Grinnell, M. Meagher, and D.B. Amos. 1961. Sex-linked incompatibility of male skin and primary tumors transplanted to isologous female mice, p. 271-294. In Genetics and Cancer. 13th Annu. Symp. Fundamental Cancer Res. University of Texas Press, Austin.
Hauschka, T.S., and B.A. Holdridge. 1962. A cytogenetic approach to the Y-linked histocompatibility antigen of mice. Ann. N.Y. Acad. Sci. 101: 12-22.
Hauschka, T.S., J.T. Mitchell, and D.J. Niederpruem. 1959.
A reliable frozen tissue bank: viability and stability of 82 neoplastic and normal cell types after prolonged storage at -78°C.
Cancer Res. 19: 643-653.
See also
PubMed.
Hellström, K.E. 1961.
Studies on the mechanism of isoantigenic variant formation in heterozygous mouse tumors. II. Behavior of H-2 antigens D and K: cytotoxic tests on mouse lymphomas.
J. Nat. Cancer Inst. 27: 1095-1105.
See also
PubMed.
Hellström, K.E. 1964. Growth inhibition of sarcoma and carcinoma cells of homozygous origin. Science 143: 477-478.
Hellström, K.E., I. Hellström, and G. Haughton. 1964. Demonstration of syngeneic preference in vitro. Nature 204: 661-664.
Herzenberg, L.A., and L.A. Herzenberg. 1961.
Association of H-2 antigens with the cell membrane fraction of mouse liver.
Proc. Natl. Acad. Sci. 47: 762-767.
See also
PubMed.
Hoecker, G., S. Counce, and P. Smith. 1954.
The antigens determined by the H-2 locus: a Rhesus-like system in the mouse.
Proc. Nat. Acad. Sci. 40: 1040-1051.
See also
PubMed.
Hoecker, G., and O. Pizarro. 1962. The histocompatibility antigens, p. 54-71. In A.P. Cristoffanini and G. Hoecker [ed.] Proceedings International Symposium Tissue Transplantation. Universidad Chile, Santiago.
Hoecker, G., O. Pizarro, and A. Ramos. 1959.
Some new antigens and histocompatibility factors in the mouse.
Tranplant. Bull. 6: 407-411.
See also
PubMed.
Kandutsch, A.A., and U. Reinert-Wenck. 1957.
Studies on a substance that promotes tumor homograft survival (the "enhancing substance"); its distribution and some properties.
J. Exp. Med. 105: 125-139.
See also
PubMed.
Kandutsch, A.A., and J.H. Stimpfling. 1963.
Partial purification of tissue isoantigens from a mouse sarcoma.
Transplantation 1: 201-216.
See also
PubMed.
Kandutsch, A.A., and J.H. Stimpfling. 1965. Properties of purified isoantigenic preparations from a mouse sarcoma, p. 1-12. In Counc. Int. Organ. Med. Sci. Symp. Immunopathol. Schwabe, Basel.
Katsh, G.F., D.W. Talmage, and S. Katsh. 1964.
Acceptance or rejection of male skins by isologous female mice; effect of injection of sperm.
Science 143: 41-42.
See also
PubMed.
Klein, E., and G. Klein. 1964.
Studies on the mechanisms of isoantigenic variant formation in heterozygous mouse tumors. III. Behavior of the H-2 antigens D and K when located in the trans position.
J. Nat. Cancers Inst. 32: 569-578.
See also
PubMed.
Klein, E., and O. Linder. 1961.
Factorial analysis of the reactivity of C57BL females against isologous male skin grafts.
Transplant. Bull. 27: 457-460.
See also
PubMed.
Linder, O.E.A. 1961.
Comparisons between survival of grafted skin, ovaries and tumors in mice across histocompatibility barriers of different strength.
J. Nat. Cancer Inst. 27: 351-373.
See also
PubMed.
Linder, O.E.A. 1963.
Skin compatibility of different CBA sublines separated from each other in the course of a varying number of generations.
Transplantation 1: 58-60.
See also
PubMed.
Little, C.C. 1914. A possible Mendelian explanation for a type of inheritance apparently non-Mendelian in nature. Science 40: 904-906.
Little, C.C. 1941. The genetics of tumor transplantation, p. 279-309. In G.D. Snell [ed.] Biology of the Laboratory Mouse. Blakiston, Philadelphia.
Little, C.C., and E.E. Tyzzer. 1916. Further studies on inheritance of susceptibility to a transplantable tumor of Japanese waltzing mice. J. Med. Res. 33: 393-425.
Manson, L.A., G.V. Foschi, and J. Palm. 1963. An association of transplantation antigens with microsomal lipoproteins of normal and malignant mouse tissues. J. Cell. Comp. Physiol. 61: 109-118.
Martinez, C., F. Shapiro, and R.A. Good. 1959.
Absence of gene interaction in mouse hybrids, revealed by studies of immunological tolerance and homotransplants.
Proc. Soc. Exp. Biol. Med. 101: 658-660.
See also
PubMed.
Mariani, T., C. Martinez, J.M. Smith, and R.A. Good. 1959. Absence of gene interaction in mouse hybrids, revealed by studies of immunological tolerance and homotransplants. Proc. Soc. Exp. Biol. Med. 101: 658-660.
Michie, D., and A. McLaren. 1958.
A proposed genetic analysis of the Eichwald-Silmser effect.
Transplant. Bull. 5: 17-18.
See also
PubMed.
Mitchison, N.A. 1956. Antigens of heterozygous tumors as material for the study of cell heredity. Proc. Roy. Phys. Soc. 25(2): 45-48.
Möller, E. 1965. Contact-induced cytotoxicity by lymphoid cells containing foreign isoantigens. Science 147: 873-879.
Old, L.J., E.A. Boyse, and E. Stockert. 1963.
Antigenic properties of experimental leukemias. I. Serological studies in vitro with spontaneous and radiation-induced leukemias.
J. Nat Cancer Inst. 31: 977-986.
See also
PubMed.
Pizarro, O., G. Joecker, P. Rubinstein, and A. Ramos. 1961.
The distribution of H-2 antigens of the mouse.
Proc. Nat. Acad. Sci. 47: 1900-1906.
See also
PubMed.
Pizarro, O., P. Rubinstein, and G. Hoecker. 1963.
Properties of histocompatibility-2 antigens from different tissues.
Guy's Hosp. Rep. 112: 392-401.
See also
PubMed.
Popp, D.M., and D.B. Amos. 1965.
An H-2 analysis of strain RFM/U mice.
Transplantation 3: 501-508.
See also
MGI.
Popp, R.A., G.E. Cosgrove, and R.D. Owen, 1958.
Genetic differences in hemoglobin as markers for bone marrow transplantation in mice.
Proc. Soc. Exp. Biol. Med. 99: 692-694.
See also
PubMed.
Popp, R.A., and G. Cudkowicz. 1965.
Independence of deficient early growth and later regression of (C57BL x 101)F2 marrow grafts in (C57BL x 101)F1 hybrid mice.
Transplantation 3: 155-160.
See also
PubMed.
Popp, D.M., and R.A. Popp. 1964. A non-H2 erythrocyte isoantigen on RFM mice. Genetics 50: 276-277. (Abstr.)
Prehn, R.T., and J.M. Main. 1958.
Number of mouse histocompatibility genes involved in skin grafting from strain BALB/cAn to strain DBA/2.
J. Nat. Cancer Inst. 20: 207-209.
See also
PubMed.
Reif, A.E., and J.M.V. Allen. 1964.
The AKR thymic antigen and its distribution in leukemias and nervous tissues.
J. Exp. Med. 120: 413-433.
See also
MGI.
Rosenfield, R.E., F.H. Allen, S.N. Swisher, and S. Kochwa. 1962.
A review of Rh serology and presentation of a new terminology.
Transfusion 2: 287-312.
See also
PubMed.
Rubin, B.A. 1960.
The determination of the H-2 genotype of CE mice.
Transplant. Bull. 26: 153-154.
See also
PubMed.
Rubinstein, P., and J.W. Ferrebee. 1964.
The H-2 phenotypes of random-bred Swiss-Webster mice.
Transplantation 2: 715-721.
See also
PubMed.
Russell, P.S., and A.P. Monaco. 1965. The Biology of Tissue Transplantation. Little, Brown, Boston. 207 p.
Shreffler, D.C. 1964.
A serologically detected variant in mouse serum: further evidence for genetic control by the histocompatibility-2 locus.
Genetics 49: 973-978.
See also
PubMed.
Shreffler, D.C. 1965.
The Ss system of the mouse — a quantitative serum protein difference genetically controlled by the H-2 antigen. P. 11-19.
In J. Palm (ed.) Isoantigens and cell interactions. Wistar Institute Press, Philadelphia.
See also
PubMed.
Shreffler, D.C., and R.D. Owen. 1963.
A serologically detected variant in mouse serum: inheritance and association with the histocompatibility-2 locus.
Genetics 48: 9-25.
See also
MGI.
Singer, M.F., M. Foster, M.L. Petras, P. Tomlin, and R.W. Sloan. 1964.
A new case of blood group inheritance in the house mouse, Mus musculus.
Genetics 50: 285-286. (Abstr.)
See also
MGI.
Snell, G.D. 1948. Methods for the study of histocompatibility genes. J. Genet. 49: 87-108.
Snell, G.D. 1951.
A fifth allele at the histocompatibility-2 locus of the mouse as determined by tumor transplantation.
J. Nat. Cancer Inst. 11: 1299-1305.
See also
PubMed.
Snell, G.D. 1952.
Enhancement and inhibition of the growth of tumor homoiotransplants by pretreatment of the hosts with various preparations of normal and tumor tissue.
J. Nat. Cancer Inst. 13: 719-729.
See also
PubMed.
Snell, G.D. 1953.
A cytosieve permitting sterile preparation of suspensions of tumor cells for transplantation.
J. Nat. Cancer Inst. 13: 1511-1515.
See also
PubMed.
Snell, G.D. 1958a.
Histocompatibility genes. I. Demonstration of weak histocompatibility differences by immunization and controlled tumor dosage.
J. Nat. Cancer Inst. 20: 787-824.
See also
PubMed.
Snell, G.D. 1958b.
Histocompatibility genes of the mouse. II. Production and analysis of isogenic resistant lines.
J. Nat. Cancer Inst. 21: 843-877.
See also
MGI.
Snell, G.D. 1959. Transplantable tumors, p. 293-345. In F. Homburger [ed.] The Physiopathology of Cancer, 2nd ed. Hoeber-Harper, New York.
Snell, G.D. 1963. The immunology of tissue transplantation, p. 323-352. In: Conceptual Advances in Immunology and Oncology. 16th Annu. Symp. Fundamental Cancer Res. (Univ. Texas, Houston). Hoeber-Harper, New York.
Snell, G.D. 1964a.
Methods for the study of histocompatibility genes and isoantigens. Introduction.
Meth. Med. Res. 10: 1-7.
See also
PubMed.
Snell, G.D. 1964b.
The terminology of tissue transplantation.
Transplantation 2: 655-657.
See also
PubMed.
Snell, G.D., and H.P. Bunker. 1964.
Histocompatibility genes of mice. IV. The position of H-3 in the fifth linkage group.
Transplantation 2: 743-751.
See also
MGI.
Snell, G.D., and H.P. Bunker. 1965.
Histocompatibility genes of mice. V. Five new histocompatibility loci identified by congenic resistant lines on a C57BL/10 background.
Transplantation 3: 235-252.
See also
MGI.
Snell, G.D., and G.F. Higgins. 1951.
Alleles at the histocompatibility-2 locus in the mouse as determined by tumor transplantation.
Genetics 36: 306-310
See also
PubMed.
Snell, G.D., G. Hoecker, D.B. Amos, and J.H. Stimplfling. 1964.
A revised nomenclature for the histocompatiblity-2 locus of the mouse.
Transplantation 2: 777-784.
See also
PubMed.
Snell, G.D., P. Smith, and F. Gabrielson. 1953.
Analysis of the histocompatibility-2 locus in the mouse.
J. Nat. Cancer Inst. 14: 457-480.
See also
PubMed.
Snell, G.D., and L.C. Stevens. 1961.
Histocompatibility genes of mice. III. H-1 and H-4, two histocompatibility loci in the first linkage group.
Immunology 4: 366-379.
See also
MGI.
Snell, G.D., N. Wheeler, and M. Aaron. 1957.
A new method of typing inbred strains of mice for histocompatibility antigens.
Transplant. Bull. 4: 18-21.
See also
PubMed.
Spencer, R.A., T.S. Hauschka, D.B. Amos, and B. Ephrussi. 1964.
Co-dominance of isoantigens in somatic hybrids of murine cells grown in vitro.
J. Nat. Cancer Inst. 33: 893-903.
See also
PubMed.
Stimpfling, J.H. 1961. The use of PVP as a developing agent in mouse hemagglutination tests. Transplant. Bull. 27: 109-111.
Stimpfling, J.H., and O. Pizarro. 1961. On the antigenic products of the H-2m allele in the laboratory mouse. Transplant. Bull. 28: 102-106.
Stimpfling, J.H., and A. Richardson. 1965.
Recombination within the histocompatibility-2 locus of the mouse.
Genetics 51: 831-846.
See also
MGI.
Welshons, W.J., and L.B. Russell. 1959.
The Y-chromosome as the bearer of male determining factors in the mouse.
Proc. Nat. Acad. Sci. 45: 560-566.
See also
MGI.
Wright, S. 1934.
An analysis of variability in number of digits in an inbred strain of guinea pigs.
Genetics 19: 506-536.
See also
PubMed.
Zaalberg, O.B. 1959.
An analysis of the Eichwald-Silmser effect.
Transplant. Bull. 6: 433-435.
See also
PubMed.
| Previous | Next |