Phenotypes associated with this allele
Allelic
Composition |
Juntm1Wag/Juntm1Wag
|
|
Genetic
Background |
either: (involves: 129S2/SvPas) or (involves: 129S2/SvPas * C57BL/6) |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Juntm1Wag mutation
(0 available);
any
Jun mutation
(12 available)
|
|
|
mortality/aging
|
|
• homozygotes die between E11.5 and E15.5; no live homozygotes are recovered after E16.5
|
|
|
• homozygotes die between E11.5 and E15.5, with frequent deaths noted at E13.5
|
liver/biliary system
|
|
• at E12.5, homozygotes often display hepatocyte apoptosis
|
|
|
• at E12.5, homozygotes often display hepatocyte necrosis
|
|
|
• at E12.5, homozygotes display absence of developing ducts
|
|
|
• homozygotes exhibit defective fetal hepatogenesis
|
|
|
• at E12.5, mutant livers display a hypoplastic epithelium with rounded, dissociated residual hepatocytes
|
homeostasis/metabolism
hematopoietic system
|
|
• homozygotes show enhanced fetal liver erythropoiesis with elevated numbers of nucleated erythroid cells, resulting in an erythroblastosis-like phenotype
|
nervous system
|
|
• at E12.5, homozygotes exhibit severe edema in the forebrain, as shown by large intercellular spaces
|
cardiovascular system
|
|
• at E12.5, 1 of 8 homozygotes exhibited severely congested vessels and signs of early liver damage
|
endocrine/exocrine glands
|
|
• at E12.5, homozygotes display absence of developing ducts
|
cellular
|
|
• at E12.5, homozygotes often display hepatocyte apoptosis
|
|
|
• at E12.5, homozygotes often display hepatocyte necrosis
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Juntm1Wag mutation
(0 available);
any
Jun mutation
(12 available)
|
|
|
cellular
|
|
• in mouse embryonic fibroblasts
|
|
|
• mouse embryonic fibroblasts and fetal liver cells produce more hydrogen peroxide than wild-type cells
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Juntm1Wag mutation
(0 available);
any
Jun mutation
(12 available)
|
|
|
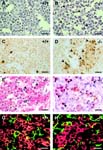
Juntm1Wag/Juntm1Wag fetuses exhibit increased apoptosis in the liver
liver/biliary system
|
|
• at E13.0, 8 of 19 mutant fetal livers show significantly increased apoptosis of both hepatoblasts and erythroid cells; however, no deregulation of apoptosis is noted at E12.5
• mutant fetal liver cells are able to reconstitute all hematopoietic compartments of lethally irradiated recipient mice, indicating that hematopoietic cell apoptosis is likely not due to a cell-autonomous defect
• in vitro, primary hepatoblasts and fibroblasts obtained from E12.5 homozygotes show a ~4-fold increase in apoptosis relative to wild-type cells
|
|
|
• in vitro, primary hepatoblasts and fibroblasts obtained from E12.5 homozygotes display a ~50% reduction in mitotic capacity as shown by [3H]thymidine incorporation
|
cardiovascular system
|
|
• at E12.5, some homozygotes display abnormal remodeling of the aortic arch arteries resulting in a right-sided aortic arch
|
|
|
• at E12.5, all (19 of 19) homozygotes exhibit a single outflow vessel arising from the right ventricle
|
|
|
• at E14.5, homozygotes display incomplete septation of the left and right ventricles
|
|
|
• at E12.5, homozygotes display a thinner right ventricular wall
|
embryo
|
|
• at E12.5, homozygotes exhibit reduced migration of neural crest cells in the outflow tract of the right ventricle
|
cellular
|
|
• at E13.0, 8 of 19 mutant fetal livers show significantly increased apoptosis of both hepatoblasts and erythroid cells; however, no deregulation of apoptosis is noted at E12.5
• mutant fetal liver cells are able to reconstitute all hematopoietic compartments of lethally irradiated recipient mice, indicating that hematopoietic cell apoptosis is likely not due to a cell-autonomous defect
• in vitro, primary hepatoblasts and fibroblasts obtained from E12.5 homozygotes show a ~4-fold increase in apoptosis relative to wild-type cells
|
|
|
• at E12.5, homozygotes exhibit reduced migration of neural crest cells in the outflow tract of the right ventricle
|
|
|
• in vitro, primary hepatoblasts and fibroblasts obtained from E12.5 homozygotes display a ~50% reduction in mitotic capacity as shown by [3H]thymidine incorporation
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Juntm1Wag mutation
(0 available);
any
Jun mutation
(12 available)
Juntm5.1(Junb)Wag mutation
(0 available);
any
Jun mutation
(12 available)
|
|
|
mortality/aging
|
|
• although mutant embryos are obtained at normal Mendelian ratios until E14.5, the frequency declines and no live mutant fetuses are recovered by E18.5
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Juntm1Wag mutation
(0 available);
any
Jun mutation
(12 available)
Tg(Junb)1598Angl mutation
(0 available)
|
|
|
mortality/aging
|
|
• only a few mutant mice survive past weaning, with the oldest one reaching 7 months of age
|
|
|
• although Mendelian ratios are recovered at E14.5-E18.5, only some viable mutant mice are born
|
|
|
• only 4% of mutant mice reach weaning age
|
vision/eye
|
|
• newborn mutant mice have open eyes but do not show any other overt defects
|
cardiovascular system
|
N |
• at E12.5, mutant fetuses exhibit normal formation of the aorta and pulmonary artery, indicating that development of the cardiac outflow tract is unaffected
|
liver/biliary system
|
N |
• at E15.5, mutant fetuses display normal liver architecture
|
cellular
|
|
• similar to primary MEFs isolated from E12.5 Juntm1(Junb)Wag homozygotes, these mutant MEFs do not enter premature senescence and show increased proliferation relative to Juntm1Wag cells, but do not proliferate as fast as wild-type cells
|



 Analysis Tools
Analysis Tools