Phenotypes associated with this allele
Allelic
Composition |
Nf1tm1Fcr/Nf1tm1Fcr
|
|
Genetic
Background |
either: (involves: 129S/SvEv) or (involves: 129S/SvEv * C57BL/6J) |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
|
|
|
mortality/aging
cellular
cardiovascular system
|
|
• show disoriented and poorly developed myocardial fibers
|
|
|
• at E13.5, have a common root of the aorta and pulmonary artery departing from the conus cordis of the right ventricle
• as the truncus proceeds cephalad, it divides into two channels, the pulmonary artery and the aorta, which are not fully separate and are joined in a common external sheath
|
|
|
• at E13.5, the atrioventricular canal is composed of loosely arranged endothelial cells that lack the typical cellular density
|
|
|
• E13.5 endocardial cushion retains a loose, myxoid appearance that is seen at E12 in wild-type, even though it merges and divides the atrioventricular canal into the left and right channels
|
|
|
• considerably larger at E13.5
|
|
|
• exhibit only a rudimentary septum near the apex that is exculsively muscular
|
|
|
• cardiac valve abnormalities at E13.5
|
|
|
• leaflets of the mitral valve remain poorly condensed at E13.5
|
liver/biliary system
|
|
• 18- to 24-hr delay in hepatic development
|
muscle
|
|
• musculature of the stomach, the three layers of the abdominal musculature, and the muscles of the shoulder girdle are thinner
|
|
|
• show disoriented and poorly developed myocardial fibers
|
|
|
• 18- to 24-hour delay in development of skeletal muscle
|
|
|
• skeletal muscle throughout the body is hypoplastic at E13.5
|
renal/urinary system
|
|
• reduced number of glomeruli at E13.5, due to developmental delay
|
|
|
• a retardation of cephalad repositioning is noted at E13.5
|
|
|
• 18- to 24-hr delay in renal development
• in the metanephros, display a retardation of cephalad repositioning at E13.5
|
vision/eye
nervous system
|
|
• seen in about 6.3% of homozygotes
|
growth/size/body
homeostasis/metabolism
immune system
|
|
• show distended lymphatics
|
respiratory system
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
|
|
|
mortality/aging
|
|
• homozygotes die by E13.5
|
homeostasis/metabolism
Allelic
Composition |
Nf1tm1Fcr/Nf1tm1.1Par
|
|
Genetic
Background |
involves: 129S/SvEv * 129S1/Sv * 129S2/SvPas * 129X1/SvJ |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1.1Par mutation
(0 available);
any
Nf1 mutation
(157 available)
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
|
|
|
mortality/aging
cardiovascular system
homeostasis/metabolism
vision/eye
pigmentation
growth/size/body
mortality/aging
|
|
• mice injected intraperitoneally with poly(I:C) at P8 to induce loss of Pten all die at 20 to 35 days of age
|
hematopoietic system
|
|
• mice injected with poly(I:C) at P8 develop myeloproliferative neoplasm with clinical manifestations of Juvenile myelomonocytic leukemia at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit anemia 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit reduced total cell numbers in bone marrow at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit decreased hemoglobin 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased platelets 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show a reduction in lymphocytes 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show decreased B-cell (CD19+) populations in PB and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show decreased T-cell (CD3e+) populations in PB and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit a modest elevation in white blood cells 2-3 weeks post poly(I:C) treatment
|
|
|
• mice injected with poly(I:C) at P8 show an elevation in granulocytes in bone marrow, blood, and spleen 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased macrophages in bone marrow, blood, and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show an elevation in monocytes in bone marrow, blood, and spleen 2-3 weeks post induction
|
|
|
• bone marrow of mice injected with poly(I:C) at P8 shows decreased hematopoietic progenitor cells, including LIN-, LIN-Sca1-1-, cKit+, and LIN-Sca1-1+cKit+, are decreased, with less apoptosis
• spleen of mice injected with poly(I:C) at P8 shows an increase in hematopoietic progenitor cells
|
|
|
• mice injected with poly(I:C) at P8 show substantial monocyte/macrophage and granulocyte infiltration in the spleen at 2-3 weeks post induction
• spleen of mice injected with poly(I:C) at P8 shows an increase in hematopoietic progenitor cells
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen size
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen weight
|
immune system
|
|
• mice injected with poly(I:C) at P8 show a reduction in lymphocytes 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show decreased B-cell (CD19+) populations in PB and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show decreased T-cell (CD3e+) populations in PB and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit a modest elevation in white blood cells 2-3 weeks post poly(I:C) treatment
|
|
|
• mice injected with poly(I:C) at P8 show an elevation in granulocytes in bone marrow, blood, and spleen 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased macrophages in bone marrow, blood, and spleen at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show an elevation in monocytes in bone marrow, blood, and spleen 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 show substantial monocyte/macrophage and granulocyte infiltration in the spleen at 2-3 weeks post induction
• spleen of mice injected with poly(I:C) at P8 shows an increase in hematopoietic progenitor cells
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen size
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen weight
|
liver/biliary system
|
|
• mice injected with poly(I:C) at P8 show substantial monocyte/macrophage infiltration in the liver at 2-3 weeks post induction
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased liver size
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased liver weight
|
neoplasm
|
|
• mice injected with poly(I:C) at P8 develop myeloproliferative neoplasm with clinical manifestations of Juvenile myelomonocytic leukemia at 2-3 weeks post induction
• recipient mice transplanted with bone marrow nucleated cells from mutants develop an indolent myelodysplastic syndrome or myeloproliferative neoplasm by 8 weeks posttransplantation
• mice injected with poly(I:C) at 6 weeks of age develop a transient myeloproliferative neoplasm after 3 weeks post induction and 1/7 transform to T-ALL
|
|
|
• mice injected with poly(I:C) at 6 weeks of age develop a transient myeloproliferative neoplasm after 3 weeks post induction and 1/7 transform to T-ALL
|
respiratory system
|
|
• mice injected with poly(I:C) at P8 show substantial monocyte/macrophage infiltration in the spleens, livers, and lungs at 2-3 weeks post induction
|
skeleton
|
|
• mice injected with poly(I:C) at P8 show increases in monocytes/macrophages and granulocytes in the bone marrow at 2-3 weeks post induction
• bone marrow of mice injected with poly(I:C) at P8 shows decreased hematopoietic progenitor cells, including LIN-, LIN-Sca1-1-, cKit+, and LIN-Sca1-1+cKit+, are decreased, with less apoptosis
|
growth/size/body
|
|
• mice injected with poly(I:C) at P8 exhibit increased liver size
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased liver weight
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen size
|
|
|
• mice injected with poly(I:C) at P8 exhibit increased spleen weight
|
mortality/aging
endocrine/exocrine glands
|
|
• medulla is overgrown compared with wild-type
|
nervous system
|
|
• peripheral ganglia are massively enlarged in newborns
|
hematopoietic system
|
|
• at 9 months of age, mutants have significantly more microglia than controls; microglia are in the optic gliomas
|
immune system
|
|
• at 9 months of age, mutants have significantly more microglia than controls; microglia are in the optic gliomas
|
nervous system
|
|
• at 9 months of age, mutants have significantly more microglia than controls; microglia are in the optic gliomas
|
|
|
• mice develop optic gliomas in the optic chiasm/nerves
• optic gliomas appear as gross thickenings and enlargements of the pre-chiasmic nerve at 9 months of age
|
|
|
• irregularly shaped and thickened astrocytes are seen in the pre-chiasmatic optic nerves
|
|
|
• retinal ganglion cell loss in the middle and peripheral region
|
|
|
• optic gliomas form in the pre-chiasmatic optic nerve/optic chiasm, with increased vascularization to the area, abnormally shaped and thickened astrocytes, increase in microglia, and axonal and myelin degeneration
• mice exhibit abnormal optic nerve axon organization
|
|
|
• the optic chiasm/nerves at 9 months of age appear as gross fusiform enlargements that result in optic gliomas
• increase in vascularization in the pre-chiasmatic optic nerve and optic chiasm (and in the optic glioma)
|
|
|
• axonal and myelin degeneration are seen in the optic nerve
|
|
|
• increase in neoplastic astrocyte proliferation in the optic glioma
|
|
|
• disruption of the myelin is seen in the optic nerve, including degeneration and hypermyelination
|
|
|
• hypermyelination is seen in the optic nerve
|
neoplasm
|
|
• mice develop optic gliomas in the optic chiasm/nerves
• optic gliomas appear as gross thickenings and enlargements of the pre-chiasmic nerve at 9 months of age
|
vision/eye
|
|
• retinal ganglion cell loss in the middle and peripheral region
|
|
|
• optic gliomas form in the pre-chiasmatic optic nerve/optic chiasm, with increased vascularization to the area, abnormally shaped and thickened astrocytes, increase in microglia, and axonal and myelin degeneration
• mice exhibit abnormal optic nerve axon organization
|
|
|
• the optic chiasm/nerves at 9 months of age appear as gross fusiform enlargements that result in optic gliomas
• increase in vascularization in the pre-chiasmatic optic nerve and optic chiasm (and in the optic glioma)
|
mortality/aging
|
|
• 2 embryos are found at later stages, after expected midgestation lethality from cardiovascular failure
|
hematopoietic system
|
|
• mice display moderate splenic enlargement and partial rescue of splenic architecture
• germinal centers are present
• splenic fibrosis is not observed
|
immune system
|
|
• mice display moderate splenic enlargement and partial rescue of splenic architecture
• germinal centers are present
• splenic fibrosis is not observed
|
nervous system
|
|
• marked enlargement of dorsal root ganglia and other neural crest derived tissues is observed
|
hematopoietic system
|
|
• normal splenic architecture is lost
|
|
|
• mice show markedly enlarged spleens due to overgrowth of granulocytic precursors
|
|
|
• germinal centers are absent
|
|
|
• splenic fibrosis is observed
|
immune system
|
|
• normal splenic architecture is lost
|
|
|
• mice show markedly enlarged spleens due to overgrowth of granulocytic precursors
|
|
|
• germinal centers are absent
|
|
|
• splenic fibrosis is observed
|
growth/size/body
|
|
• mice show markedly enlarged spleens due to overgrowth of granulocytic precursors
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
Tg(GFAP-cre)#Gtm mutation
(0 available)
Tsc1tm1Djk mutation
(2 available);
any
Tsc1 mutation
(69 available)
|
|
|
mortality/aging
|
|
• most mice die before 3 months of age
|
neoplasm
|
N |
• mice do not develop gliomas
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
Tg(GFAP-cre)#Gtm mutation
(0 available)
|
|
|
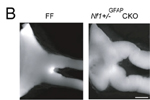
Nf1tm1Fcr/Nf1+ Tg(GFAP-cre)#Gtm/0 mice develop prechiasmatic optic nerve and chiasmal gliomas
nervous system
|
|
• cell proliferation in the optic nerve is increased compared to in wild-type mice
|
|
|
• kinking of the prechiasmatic optic nerves
|
neoplasm
vision/eye
|
|
• kinking of the prechiasmatic optic nerves
|
cellular
|
|
• cell proliferation in the optic nerve is increased compared to in wild-type mice
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
Tg(GFAP-cre)25Mes mutation
(2 available)
Trp53tm1Elee mutation
(0 available);
any
Trp53 mutation
(232 available)
|
|
|
mortality/aging
|
|
• all mice die prior to 200 days
|
neoplasm
|
|
• 70% of gliomas exhibit necrosis, a feature of human glioblastoma multiforme
• tumors are relatively homogeneous histological and in lineage marker expression
|
|
|
• all mice develop astrocytic gliomas with shorter latency than observed in Trp53tm1Elee/Trp53tm1Tyj Tg(GFAP-cre)25Mes or Trp53tm1Elee/ Trp53tm1Elee Tg(GFAP-cre)25Mes mice
|
nervous system
|
|
• 70% of gliomas exhibit necrosis, a feature of human glioblastoma multiforme
• tumors are relatively homogeneous histological and in lineage marker expression
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gt(ROSA)26Sortm1Fia mutation
(0 available);
any
Gt(ROSA)26Sor mutation
(942 available)
Hprt1tm1(CMV-cre)Brd mutation
(0 available);
any
Hprt1 mutation
(1273 available)
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
|
|
|
mortality/aging
 |
• some mice (5/208) survive to birth while all Nf1-null homozygotes expressing either Gt(ROSA)26Sortm1Fia or Hprt1tm1(CMV-cre)Brd die prior to birth
|
nervous system
 |
• peripheral ganglia are massively enlarged in newborns
|
 |
• enormous paraspinal masses arise from the dorsal root ganglia
|
endocrine/exocrine glands
 |
• medulla is overgrown compared with wild-type, with cortical effacement and tumor-like medullary protrusion
|
homeostasis/metabolism
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gt(ROSA)26Sortm1Fia mutation
(0 available);
any
Gt(ROSA)26Sor mutation
(942 available)
Hprt1tm1(CMV-cre)Brd mutation
(0 available);
any
Hprt1 mutation
(1273 available)
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
|
|
|
nervous system
 |
• peripheral ganglia are massively enlarged in newborns
|
nervous system
|
|
• develop sarcomas and brain tumors as early as 15 weeks of age
|
mortality/aging
|
|
• mice that develop malignant tumors die by 26 weeks of age
|
neoplasm
|
|
• 11 of 19 (57.9%) mice develop malignant tumors and die by 26 weeks of age
|
|
|
• develop sarcomas and brain tumors as early as 15 weeks of age
|
|
|
• develop sarcomas and brain tumors as early as 15 weeks of age
|
mortality/aging
|
|
• die as early as 3 weeks of age
|
neoplasm
|
|
• develop tumors, primarily lymphomas
|
Allelic
Composition |
Nf1tm1Fcr/Nf1+
Trp53tm1Tyj/Trp53+
|
|
Genetic
Background |
involves: 129S/SvEv * 129S2/SvPas * C57BL/6 |
|
|
|
mortality/aging
|
|
• cis-double heterozygotes died at 15 weeks and trans-double heterozygotes died at 25 weeks
|
neoplasm
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
|
|
• both cis- and trans-double heterozygotes developed sarcomas
|
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
integument
|
|
• developed in cis-double heterozygotes
|
muscle
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
nervous system
|
|
• developed in cis-double heterozygotes
|
|
|
• developed in cis-double heterozygotes
|
Allelic
Composition |
Egfrwa2/Egfr+
Nf1tm1Fcr/Nf1+
Trp53tm1Brd/Trp53+
|
|
Genetic
Background |
involves: 129S/SvEv * 129S7/SvEvBrd * C57BL/6 * C57BL/6JEi * C3H/HeSnJ |
|
|
|
neoplasm
|
|
• only 1 of 17 (5.9%) mice develop a tumor, a spindle cell tumor compared to 57.9% of double heterozygous Nf1 and Trp53 mutants; this mouse was sacrificed at 21 weeks of age
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Nf1tm1Fcr mutation
(3 available);
any
Nf1 mutation
(157 available)
Tg(Cnp-EGFR)10Nrat mutation
(0 available)
|
|
|
nervous system
|
|
• mutants exhibit similar diffuse nerve enlargement as in single Tg(Cnp-EGFR)10Nrat mice
|
|
|
• saphenous nerves are larger at 4 months of age compared to wild-type mice but similar in size to those in single Tg(Cnp-EGFR)10Nrat mice
|
neoplasm
|
|
• no tumors are detected compared to single Tg(Cnp-EGFR)10Nrat mice
|



 Analysis Tools
Analysis Tools
