Phenotypes associated with this allele
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
mortality/aging
|
|
• homozygotes survive to term but die shortly after birth of cyanosis caused by right ventricular outflow tract obstruction
• homozygotes delivered by Cesarian section die shortly after delivery; occasionally, pups survive up to 5 hrs after birth
|
cardiovascular system
|
|
• mutant hearts display a slight increase in trabeculation of the right ventricle relative to wild-type or heterozygous hearts
|
|
|
• at E16.5 and at term, homozygotes exhibit a consistent conus defect, manifest as an enlarged right ventricular outflow tract
• at term, the enlarged right ventricular cavity is abnormally filled with intraventicular septae that divide the tract into separate, interconnected or blind-ended chambers
|
|
|
• at E16.5 and at term, homozygotes exhibit a consistent conus defect, manifest as an enlarged right ventricular outflow tract
|
|
|
• newborns display an obstruction of the subpulmonary right ventricular outflow tract from the heart to the newly ventilated lungs resulting in failure of pulmonary gas exchange
|
|
|
• PDGF-induced vascular smooth muscle cell proliferation in low serum media is ablated
|
respiratory system
|
|
• homozygous mutant pups exhibit labored breathing and swollen abdomens and stomach
|
homeostasis/metabolism
muscle
|
|
• mutant hearts display a slight increase in trabeculation of the right ventricle relative to wild-type or heterozygous hearts
|
|
|
• PDGF-induced vascular smooth muscle cell proliferation in low serum media is ablated
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
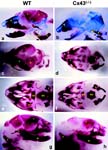
E16.5 and E18.5 Gja1tm1Kdr/Gja1tm1Kdr skulls are hypomineralized
skeleton
|
|
• cultured calvaria osteoblasts show a decrease in alkaline phosphatase activity, indicating abnormal differentiation
|
|
|
• gap junctional communication is impaired in osteoblasts as indicated by minimal cell to cell transmission of calcein
• calvarial osteoblasts grown in mineralization medium show a reduced capacity to form mineralized nodules
|
|
|
• cranial bones originating from migratory neural crest cells are hypoplastic, leaving an open foramen at birth
|
|
|
• mutants have a more prominent cartilaginous anlage of the occipital bone
|
|
|
• open parietal foramen
• thinner, brittle parietal bones with reduced diploic space
|
|
|
• lower incisors are not as prominent at birth as in wild-type
|
|
|
• incisor teeth are not visible at E18.5 as they are in wild-type, and although they are visible at birth, they are less prominent
|
|
|
• smaller at E18.5 and at birth
|
|
|
• maxillary bones are slightly smaller
|
|
|
• jugal bone of the zygomatic arch is shorter, resulting in a misshapen zygomatic arch
|
|
|
• the thoracic cage is more brittle at E15.5
|
|
|
• ribs appear jagged-shaped and slightly thinner at E15.5
|
|
|
• delayed ossification of the calvarium bones, clavicles, ribs, vertebrae, and limbs
|
|
|
• bone segments formed by endochondral ossification, including pterygoid, alisphenoid, and basisphenoid, show delayed ossification
• endochondral ossification of the axial skeleton is delayed by 1-2 days, but is normal by birth
|
|
|
• intramembranous ossification of the cranial vault and the clavicle is delayed
|
craniofacial
|
|
• cranial bones originating from migratory neural crest cells are hypoplastic, leaving an open foramen at birth
|
|
|
• mutants have a more prominent cartilaginous anlage of the occipital bone
|
|
|
• open parietal foramen
• thinner, brittle parietal bones with reduced diploic space
|
|
|
• lower incisors are not as prominent at birth as in wild-type
|
|
|
• incisor teeth are not visible at E18.5 as they are in wild-type, and although they are visible at birth, they are less prominent
|
|
|
• smaller at E18.5 and at birth
|
|
|
• maxillary bones are slightly smaller
|
|
|
• jugal bone of the zygomatic arch is shorter, resulting in a misshapen zygomatic arch
|
|
|
• slightly smaller and pointed snout
|
|
|
• slightly smaller and pointed snout
|
hearing/vestibular/ear
|
|
• smaller and thinner at birth
|
cellular
|
|
• cultured calvaria osteoblasts show a decrease in alkaline phosphatase activity, indicating abnormal differentiation
|
|
|
• gap junctional communication is impaired in osteoblasts as indicated by minimal cell to cell transmission of calcein
• calvarial osteoblasts grown in mineralization medium show a reduced capacity to form mineralized nodules
|
growth/size/body
|
|
• lower incisors are not as prominent at birth as in wild-type
|
|
|
• incisor teeth are not visible at E18.5 as they are in wild-type, and although they are visible at birth, they are less prominent
|
|
|
• slightly smaller and pointed snout
|
|
|
• slightly smaller and pointed snout
|
respiratory system
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
behavior/neurological
|
|
• newborn homozygotes fail to feed and are cast aside by their mothers
|
cardiovascular system
|
|
• attempts to record spontaneous or paced electrical activity in neonatal homozygous mutant hearts were unsuccessful, suggesting severe functional defects
|
Allelic
Composition |
Gja1tm1Kdr/Gja1tm1Kdr
|
|
Genetic
Background |
involves: 129S1/Sv * 129X1/SvJ * C57BL/6 * CD-1 |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
endocrine/exocrine glands
 |
• cultured neonatal ovaries failed to develop follicles with multuple layers of granulosa cells
|
reproductive system
|
|
• reduced number of germ cells in the fetal gonads of both sexes
|
 |
• cultured neonatal ovaries failed to develop follicles with multuple layers of granulosa cells
|
cellular
|
|
• reduced number of germ cells in the fetal gonads of both sexes
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
reproductive system
 |
• testes grafted from mutant fetuses under the kidney capsules of adult males and allowed to grow for 3 weeks contain at least 90% fewer germ cells
|
cellular
 |
• testes grafted from mutant fetuses under the kidney capsules of adult males and allowed to grow for 3 weeks contain at least 90% fewer germ cells
|
Allelic
Composition |
Gja1tm1Kdr/Gja1+
|
|
Genetic
Background |
involves: 129S1/Sv * 129X1/SvJ * C57BL/6 |
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
cardiovascular system
|
|
• 22 of 25 show various defects in patterning of the coronary arteries, including abnormal origin and course of the main coronary artery stems, multiple accessory coronary arteries, and dual septal-conal branches
|
|
|
• in neonatal heterozygotes, ventricular epicardial conduction of paced beats is 30% slower than in wild-type neonates
• in adult (6-9 mo-old) heterozygotes, ventricular epicardial conduction is 44% slower than in wild-type hearts; no differences are observed in refractory periods
• no apparent differences in wall thickness or the orientation or fiber curvature in the three muscle layers of the left ventricle are observed
|
|
|
• standard 3-lead surface ECGs from adult heterozygotes indicate a significantly prolonged QRS interval relative to wild-type mice (13.41.8 ms vs 11.51.4 ms, respectively)
• no differences are observed in spontaneous heart rates either during isolated heart studies in vitro or by electrocardiography
• no differences are noted in atrioventricular conduction times in isolated neonatal heterozygous hearts, or in PQ intervals in adult ECG studies
• whole-cell recordings display no differences in any action potential parameters in ventricular myocytes isolated from neonatal heterozygous and wild-type mice
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1.1Kwi mutation
(0 available);
any
Gja1 mutation
(59 available)
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
|
|
|
mortality/aging
|
|
• homozygotes die within 1 day after birth
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
Gja1tm1Kwi mutation
(1 available);
any
Gja1 mutation
(59 available)
Tg(Tek-cre)5326Sato mutation
(0 available)
|
|
|
normal phenotype
|
|
• mutants lacking Gja1 in endothelium are viable, fertile, and show no abnormalities in the heart, brain, retina, pancreas, liver, kidney, spleen, testis, or blood pressure
|
|
|
| Find Mice |
Using the International Mouse Strain Resource (IMSR)
Mouse lines carrying:
Gja1tm1Kdr mutation
(1 available);
any
Gja1 mutation
(59 available)
Tg(CMV-Gja1)BClo mutation
(0 available)
|
|
|
mortality/aging
cardiovascular system
|
|
• exhibit a network of fiber-like projections over the surface of the heart that are not seen in controls
|
|
|
• conotruncal enlargement of the right ventricle with multiple bulges
|
|
|
• blood flow into the right ventricular tract shows varying degrees of obstruction, with the most severe obstruction leading to backflow of the injected dye into the atrial chambers
|
muscle
|
|
• conotruncal enlargement of the right ventricle with multiple bulges
|
growth/size/body
|
|
• conotruncal enlargement of the right ventricle with multiple bulges
|



 Analysis Tools
Analysis Tools

